Introduction
Lung cancer is the leading cause of cancer-related mortality worldwide. According to 2018 data, approximately 2.09 million new cases and 1.76 million deaths were reported [1]. Among all tumours, lung cancer stands out due to the availability of molecularly targeted therapeutic approaches [2].
Computed tomography-guided transthoracic lung biopsy (CTTB) is a minimally invasive procedure that provides tissue samples for histopathological evaluation and other diagnostic tests. However, this technique is associated with certain complications [3]. The present study aims to evaluate the relationship between these complications and both the technical aspects of CTTB and lesion characteristics.
Material and methods
This study was conducted through a retrospective analysis of data from patients who underwent CTTB between 2015 and 2020. This research study was conducted on humans according to the Helsinki Declaration of 1975, as revised in 2013. Approval for the study was obtained from the local research Ethics Committee with the number 2021/3137. Biopsies performed without traversing the lung parenchyma were excluded, and a total of 510 patients were included.
Prior to biopsy, all patients underwent non-contrast, sequential axial CT scans with a slice thickness of 3 mm to localise the lesion and guide the procedure. Tube voltage ranged from 100 to 120 kV, and tube current from 100 to 200 mAs.
Patients were categorised according to technical approach and needle gauge as follows:
18G NCx group (n = 101): pleura crossed with an 18G automated biopsy needle alone, without a coaxial system (NCx);
17G Cx group (n = 98): pleura crossed using a 17G coaxial (Cx) needle, with an 18G automated biopsy needle advanced through it;
19G Cx group (n = 171): pleura crossed using a 19G Cx needle, with a 20G automated biopsy needle advanced through it;
17G Cx + OBC group (n = 49): similar to the 17G Cx group, but with autologous blood clot (OBC) injected into the biopsy tract at the end of the procedure;
19G Cx + OBC group (n = 91): similar to the 19G Cx group, with OBC injected into the tract post-procedure.
Lesion size was measured as the maximum axial diameter and classified as < 20 mm or ≥ 20 mm. Lesions were also categorised by lobar location. The pleura-to-target distance was measured along the biopsy tract and grouped as ≤ 20 mm, 21-39 mm, or ≥ 40 mm. For each patient, the number of tissue cores and pleural passes was recorded and classified as ≤ 2 or ≥ 3.
The most direct and shortest path to the target lesion was selected to maximise tissue yield while avoiding fissures, large bullae, and major vessels. Perilesional emphysema was assessed based on the criteria described by Goddard et al. [4], including pruning of small vascular branches, reduced branching of the vascular tree, distortion of vascular structures within low-attenuation areas, and enlargement of the main pulmonary arteries. Needle tracts were classified as with or without emphysema. Breath-hold techniques were not used. To prevent air aspiration during needle withdrawal, the coaxial introducer was sealed. Tru-cut biopsies were performed until adequate tissue was obtained. In OBC groups, an autologous clot was injected through the coaxial needle along the biopsy tract until the pleural margin was reached.
Procedural complications such as haemoptysis, pneumothorax (Ptx), type 1 parenchymal contusion (T1PC), haemothorax, and air embolism were assessed on post-procedure CT. Following biopsy, patients were monitored in a supine position with the punctured lung dependent. Vital signs were monitored regularly for 4-6 hours, followed by chest radiography.
Ptx was classified according to the maximum depth of pulmonary surface retraction on post-biopsy CT: 1) ≤ 20 mm, mild Ptx; and 2) > 20 mm, significant Ptx. Management was based on current evidence [5-7]. Mild Ptx was managed conservatively with clinical and vital sign monitoring, and chest radiographs were obtained 6-8 hours after biopsy. Patients who remained asymptomatic and showed no progression of Ptx were considered safe for discharge. Symptomatic patients were monitored with chest radiographs for 24 hours to exclude progression. Significant Ptx was treated with urgent manual aspiration via the outer cannula of the coaxial system or with an intercostal catheter (8-10 F). The application was carried out in accordance with the literature [5]. If respiratory distress worsened or aspiration exceeded 670 ml without improvement, placement of a large-bore (16-22 F) intercostal chest tube for underwater drainage was required.
Parenchymal contusion was classified as T1PC present or absent. T1PC was not evaluated in patients who received OBC.
Potential risk factors were analysed as follows: patient-related (age, sex, emphysema along the biopsy tract), lesion-related (lesion size, location by lobe), and technical (length of aerated lung traversed, needle gauge, coaxial vs. non-coaxial approach, use of OBC, number of pleural passes, number of tissue samples). The incidence of complications – including Ptx, chest tube-requiring Ptx, T1PC, haemoptysis, haemothorax, and haemothorax with air embolism – was calculated. Statistical comparisons were performed to identify risk factors associated with Ptx and chest tube placement.
Statistical analysis
Descriptive statistics were presented as mean ± standard deviation (SD) for continuous variables and as frequencies with percentages for categorical variables. The effects of age, sex, emphysema along the needle tract, lesion size, length of aerated lung traversed, lesion location, lesion approach, and number of samples obtained on Ptx, chest tube-requiring Ptx, and T1PC were analysed using Pearson’s χ2 test. The impact of the biopsy method on the occurrence of T1PC, Ptx, and chest tube-requiring Ptx was also assessed with univariate analysis using Pearson’s χ2 test. In addition, the effect of the number of pleural passes on Ptx, chest tube-requiring Ptx, and T1PC was evaluated by univariate analysis with Fisher’s exact test. Multivariate analysis with logistic regression was performed to identify independent risk factors for Ptx and chest tube-requiring Ptx. The association between patients who developed T1PC and those who developed Ptx was examined using Pearson’s χ2 test. Finally, the effects of needle gauge and T1PC on the development of Ptx were analysed using binomial logistic regression.
A p-value of < 0.05 was considered statistically significant for all analyses.
Results
Among the 510 patients included in the study, 340 were male and 170 were female. A total of 195 patients were ≤ 60 years old, while 315 were older than 60 years. Descriptive characteristics of the lesions, procedural techniques, pathological findings, and complication rates are summarized in a single table (Table 1).
Table 1
Descriptive characteristics, biopsy techniques, and complications
Pneumothorax analysis
No statistically significant differences in Ptx incidence were observed with respect to age, lesion size, lesion approach, or the length of aerated lung traversed (p > 0.05) (Table 2).
Table 2
Univariate analysis of risk factors for pneumothorax
| Variable | Pneumothorax | ||
|---|---|---|---|
| Yes | No | p-value | |
| Age | |||
| ≤ 60 | 40 (20.5%) | 155 (79.4%) | > 0.05* |
| > 60 | 84 (26.6%) | 231(73.3%) | |
| Sex | |||
| Male | 93 (27.3%) | 247 (72.6%) | < 0.05* |
| Female | 31 (18.2%) | 139 (81.7%) | |
| Emphysema along the needle tract | |||
| Absent | 83 (20.1%) | 330 (79.9%) | < 0.001* |
| Present | 41 (42.2%) | 56 (57.7%) | |
| Lesion size | |||
| ≤ 20 mm | 29 (19.5%) | 119 (80.4%) | > 0.05* |
| > 20mm | 95 (26.2%) | 267 (73.7%) | |
| Length of aerated lung traversed | |||
| ≤ 20 mm | 51 (29.8%) | 120 (70.1%) | > 0.05* |
| 21-39 mm | 51 (19.9%) | 205 (80.0%) | |
| ≥ 40 mm | 22 (26.5%) | 61 (73.4%) | |
| Lesion location | |||
| Upper lobe | 60 (20.9%) | 226 (79.0%) | < 0.05* |
| Lower lobe | 57 (27.4%) | 151 (72.6%) | |
| Middle lobe | 7 (43.7%) | 9 (56.2%) | |
| Lesion approach | |||
| Anterior | 20 (16.6%) | 100 (83.3%) | 0.05* |
| Posterior | 79 (25.5%) | 230 (74.4%) | |
| Lateral | 25 (30.8%) | 56 (69.1%) | |
| Biopsy method | |||
| 18G NCx | 39 (38.6%) | 62 (61.3%) | < 0.001* |
| 17G Cx | 19 (19.3%) | 79 (80.6%) | |
| 19G Cx | 49 (28.6%) | 122 (71.3%) | |
| 17G CxOBC | 5 (10.2%) | 44 (89.8%) | |
| 19G CxOBC | 12 (13.1%) | 79 (86.8%) | |
| Autologous blood clot (OBC) | |||
| Var | 17 (12.1%) | 123 (87.9%) | < 0.001* |
| Yok | 107 (28.9%) | 263 (71.1%) | |
| Number of tissue samples | |||
| ≤ 2 | 93 (22.2%) | 326 (77.8%) | < 0.05* |
| ≥ 3 | 31 (34.0%) | 60 (65.9%) | |
| Number of pleural passes | |||
| ≤ 2 | 119 (23.6%) | 385 (76.3%) | < 0.05** |
| ≥ 3 | 5 (83.3%) | 1 (16.6%) | |
| Type 1 parenchymal contusion (T1PC) | |||
| Present | 39(18.2%) | 175 (81.8%) | <0.001* |
| Absent | 68 (43.6%) | 88 (56.4%) | |
By sex, Ptx was more common in male patients, and the difference was statistically significant (p < 0.05). The presence of emphysema along the needle tract was also associated with a significantly higher risk of Ptx compared to its absence (p < 0.001) (Table 2).
Lesion location analysis showed that Ptx incidence was significantly higher in middle lobe lesions compared with upper and lower lobe lesions (p < 0.05) (Table 2).
When biopsy techniques were compared, the 18G NCx method had a higher frequency of Ptx compared with other methods, whereas techniques using OBC demonstrated lower Ptx rates. Regarding needle size, biopsies performed with a 17G Cx needle (17G Cx and 17G CxOBC) had lower Ptx incidence compared with those performed with a 19G Cx needle (19G Cx and 19G CxOBC). The difference among methods was statistically significant (p < 0.001) (Table 2).
Patients from whom ≥ 3 samples were obtained had a significantly higher incidence of Ptx compared to those with ≤ 2 samples (p < 0.05). Similarly, ≥ 3 pleural passes was associated with a significantly higher Ptx incidence compared with ≤ 2 passes (p < 0.05) (Table 2).
Finally, the presence of T1PC was associated with a statistically significant reduction in Ptx incidence (p < 0.001) (Table 2).
Chest tube-requiring pneumothorax analysis
No statistically significant differences were observed in the incidence of chest tube-requiring Ptx with respect to age, sex, length of aerated lung traversed, presence of emphysema along the needle tract, lesion size, lesion location, lesion approach, or T1PC status (p > 0.05) (Table 3).
Table 3
Univariate analysis of risk factors for chest tube-requiring pneumothorax
| Variable | Chest tube-requiring pneumothorax | ||
|---|---|---|---|
| Yes | No | p-value | |
| Age (years) | |||
| ≤ 60 | 12 (6.2%) | 183 (93.8%) | > 0.05* |
| > 60 | 21 (6.7%) | 294 (93.3%) | |
| Sex | |||
| Male | 7 (4.1%) | 163 (95.9%) | > 0.05* |
| Female | 26 (7.6%) | 314 (92.4%) | |
| Emphysema along the needle tract | |||
| Absent | 26 (6.3%) | 387 (93.7%) | > 0.05* |
| Present | 7 (7.2%) | 90 (92.8%) | |
| Lesion size | |||
| ≤ 20 mm | 10 (6.8%) | 138 (93.2%) | > 0.05* |
| > 20 mm | 23 (6.4%) | 339 (93.6%) | |
| Length of aerated lung traversed | |||
| ≤ 20 mm | 6 (3.5%) | 165 (96.5%) | > 0.05* |
| 21-39 mm | 18 (7.0%) | 238 (93.0%) | |
| ≥ 40 mm | 9 (10.8%) | 74 (89.2%) | |
| Location | |||
| Upper lobe | 20 (6.9%) | 266 (93.0%) | > 0.05* |
| Lower lobe | 12 (5.7%) | 196 (94.2%) | |
| Middle lobe | 1 (6.2%) | 15 (93.7%) | |
| Lesion approach | |||
| Anterior | 8 (6.6%) | 112 (93.3%) | > 0.05* |
| Posterior | 22 (7.1%) | 287 (92.8%) | |
| Lateral | 3 (3.7%) | 78 (96.3%) | |
| Biopsy method | |||
| 18G NCx | 4 (3.9%) | 97 (96.0%) | < 0.05* |
| 17G Cx | 3 (3.0%) | 95 (96.9%) | |
| 19G Cx | 19 (11.1%) | 152 (88.9%) | |
| 17G CxOBC | 0 (0%) | 49 (100.0%) | |
| 19G CxOBC | 7 (7.7%) | 84 (92.3%) | |
| Autologous blood clot (OBC) | |||
| Var | 7 (5.0%) | 133 (95.0%) | > 0.05* |
| Yok | 26 (7.0%) | 344 (93.0%) | |
| Number of tissue samples | |||
| ≤ 2 | 19 (4.5%) | 400 (95.5%) | < 0.05* |
| ≥ 3 | 14 (15.4%) | 77 (84.6%) | |
| Number of pleural passes | |||
| ≤ 2 | 29 (5.8%) | 475 (94.2%) | < 0.001** |
| ≥ 3 | 4 (66.7%) | 2 (33.3%) | |
| Type 1 parenchymal contusion (T1PC) | |||
| Present | 16 (7.5%) | 198 (92.5%) | > 0.05* |
| Absent | 10 (6.4%) | 146 (93.6%) | |
Comparison of biopsy techniques showed significant differences: procedures performed with a 17G Cx system had lower chest tube-requiring Ptx rates compared with those performed with a 19G Cx system (p < 0.05) (Table 3). The use of OBC was not significantly associated with chest tube-requiring Ptx (p > 0.05) (Table 3).
Patients with ≥ 3 tissue samples had a significantly higher incidence of chest tube-requiring Ptx than those with ≤ 2 samples (p < 0.001). Likewise, ≥ 3 pleural passes was associated with significantly higher rates of chest tube-requiring Ptx compared with ≤ 2 passes (p < 0.001) (Table 3).
Multivariate logistic regression revealed that OBC use was significantly associated with lower Ptx incidence (p < 0.05, OR: 0.39, CI: 0.21-0.74). Use of the 18G NCx system was significantly associated with higher Ptx incidence compared with the 17G Cx system (p < 0.001, OR: 4.08, CI: 2.01-8.28). Similarly, use of the 19G Cx system was associated with higher Ptx incidence than the 17G Cx system (p < 0.05, OR: 2.92, CI: 1.52-5.60). In addition, lateral lesion approach, presence of emphysema along the needle tract, ≥ 3 pleural passes, and ≥ 3 tissue samples were all identified as significant risk factors for Ptx development (p < 0.05) (Table 4).
Table 4
Multivariate analysis results for identifying independent risk factors of pneumothorax and chest tube-requiring pneumothorax
For chest tube-requiring Ptx, multivariate models showed that ≥ 3 pleural passes and ≥ 3 samples were independently associated with higher risk (p < 0.05, OR: 28.84, CI: 2.80-296.11; and p < 0.001, OR: 4.26, CI: 1.82-9.94, respectively). Use of the 19G Cx system was also significantly associated with increased chest tube-requiring Ptx compared to the 17G Cx system (p < 0.05, OR: 6.01, CI: 1.71-21.05). Use of the 18G NCx needle, however, was not significantly associated with chest tube-requiring Ptx compared with the 17G Cx system (p > 0.05) (Table 4).
Type 1 parenchymal contusion analysis
No statistically significant differences in T1PC incidence were found according to age, sex, presence of emphysema along the needle tract, lesion size, lesion approach, or length of aerated lung traversed (p > 0.05). Although ≥ 3 pleural passes was associated with a higher T1PC rate compared with ≤ 2 passes, the difference was not statistically significant (p > 0.05) (Table 5).
Table 5
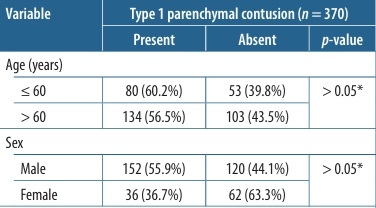
Univariate analysis results for identifying risk factors of type 1 parenchymal contusion complication
| Variable | Type 1 parenchymal contusion (n = 370) | ||
|---|---|---|---|
| Present | Absent | p-value | |
| Age (years) | |||
| ≤ 60 | 80 (60.2%) | 53 (39.8%) | > 0.05* |
| > 60 | 134 (56.5%) | 103 (43.5%) | |
| Sex | |||
| Male | 152 (55.9%) | 120 (44.1%) | > 0.05* |
| Female | 36 (36.7%) | 62 (63.3%) | |
| Emphysema along the needle tract | |||
| Absent | 168 (58.3%) | 120 (41.7%) | > 0.05* |
| Present | 46 (56.1%) | 36 (43.9%) | |
| Lesion size | |||
| ≤ 20 mm | 59 (60.2%) | 39 (39.8%) | > 0.05* |
| > 20 mm | 155 (57.0%) | 117 (43.0%) | |
| Traversed aerated lung parenchyma length | |||
| ≤ 20 mm | 78 (55.3%) | 63 (44.7%) | > 0.05* |
| 21-39 mm | 107 (58.2%) | 77 (41.8%) | |
| ≥ 40 mm | 29 (64.4%) | 16 (35.6%) | |
| Location | |||
| Upper lobe | 113 (57.4%) | 84 (42.6%) | > 0.05* |
| Lower lobe | 95 (59.7%) | 64 (40.3%) | |
| Middle lobe | 6 (42.9%) | 8 (57.1%) | |
| Anterior | 53 (61.6%) | 33 (38.4%) | > 0.05* |
| Posterior | 132 (59.2%) | 91 (40.8%) | |
| Lateral | 29 (47.5%) | 32 (52.5%) | |
| Biopsy method | |||
| 18G NCx | 49 (48.5%) | 52 (51.5%) | < 0.001* |
| 17G Cx | 75 (76.5%) | 23 (23.5%) | |
| 19G Cx | 81 (52.6%) | 90 (47.4%) | |
| Number of tissue samples | |||
| ≤ 2 | 19 (4.5%) | 400 (95.5%) | < 0.05* |
| ≥ 3 | 14 (15.4%) | 77 (84.6%) | |
| Number of pleural passes | |||
| ≤ 2 | 211 (57.7%) | 155 (42.3%) | > 0.05** |
| ≥ 3 | 3 (75.0%) | 1 (25.0%) | |
Comparison of biopsy techniques showed that procedures performed with a 17G Cx system had a significantly higher incidence of T1PC compared with both 18G NCx and 19G Cx systems (p < 0.001) (Table 5). As noted previously, methods involving OBC were excluded from T1PC analysis.
Patients from whom ≥ 3 samples were obtained had a significantly higher incidence of T1PC compared to those with ≤ 2 samples (p < 0.05) (Table 5).
Multivariate analysis demonstrated that use of the 18G NCx system was associated with a significant reduction in T1PC compared with the 17G Cx system (p < 0.001, OR: 0.33, CI: 0.18-0.66). Similarly, use of the 19G Cx system resulted in a significant reduction in T1PC compared with the 17G Cx system (p < 0.001, OR: 0.35, CI: 0.20-0.63). In addition, the absence of Ptx was associated with an increased likelihood of T1PC (p < 0.001, OR: 0.31, CI: 0.19-0.50) (Table 6).
Table 6
Multivariate analysis results for identifying independent risk factors of type 1 parenchymal contusion complication and its association with pneumothorax
| Variable | Reference variable | p-value* | Odds ratio | 95% CI | |
|---|---|---|---|---|---|
| Contusion | 18G NCx | 17G Cx | < 0.001 | 0.33 | 0.18-0.63 |
| 19G Cx | 17G Cx | < 0.001 | 0.35 | 0.20-0.63 | |
| Pneumothorax present | Pneumothorax absent | < 0.001 | 0.31 | 0.19-0.50 |
Discussion
In CT-guided tru-cut lung biopsies, Ptx, chest tube-requiring Ptx, and T1PC can occur. These complications will be discussed separately below.
Pneumothorax
Ptx is one of the most common complications of CTTB. The increased use of screening CT in high-risk populations has led to a higher detection rate of nodules and masses. Consequently, the number of CTTBs performed both in clinical practice and research is likely to increase. Minimising the incidence of Ptx after biopsy remains a challenge. Identifying individual and technical factors that may contribute to increased risk of Ptx and subsequent chest tube placement is crucial for understanding the underlying causes and implementing corrective measures. The choice of Cx or NCx techniques, different needle calibres, and the use of on-site coaxial biopsy (OBC) are generally left to the discretion of the operator. Regardless of the technique used, the same patient- and lesion-related risk factors are shared.
In the current study, comparison according to age showed a higher incidence of Ptx in patients over 60 years old; however, this difference was not statistically significant (p > 0.05) (Table 2). Nour-Eldin et al. [8] structured age groups similarly to our study and reported, in agreement with our findings, that patient age was not associated with the frequency of Ptx. Yeow et al. [9] found that age was a significant risk factor for Ptx in univariate analysis, but multivariate regression did not demonstrate a significant association between patient age and Ptx development. Similarly, Hiraki et al. [10] reported no statistically significant relationship between patient age and Ptx incidence, consistent with our data. Conversely, studies by Kuban et al. [7], Geraghty et al. [11], and Kim et al. [12] have indicated that advanced age is associated with an increased incidence of Ptx.
When analysed by sex in our study, the incidence of Ptx was significantly higher in males (p < 0.05) (Table 2). The literature presents mixed findings regarding sex differences. Laurent et al. [13] reported a higher incidence of Ptx in males compared to females in patients with lesion diameters ≤ 20 mm and > 20 mm. Kim et al. [12] also found significantly higher Ptx rates in males (p < 0.001). However, Nour-Eldin et al. [8] reported no statistically significant difference in Ptx incidence between male and female patients undergoing Cx or NCx biopsies. Hiraki et al. [10] similarly found no significant sex-based difference (p > 0.05). We did not identify any studies reporting higher Ptx rates in females.
The presence of emphysema has been reported as a statistically significant risk factor for the development of Ptx in several studies [7,8,11,12,14-16]. Our results are consistent with these observations. We found a significantly higher incidence of Ptx in patients with emphysema along the needle tract. Both univariate and multivariate analyses demonstrated a significant association between Ptx development and the presence of emphysema along the needle pathway (p < 0.001) (Table 2). Malone et al. [16] reported a significantly higher incidence of Ptx in patients with emphysema (p < 0.05). Graffy et al. [15] identified moderate-to-severe emphysema as an independent risk factor for Ptx development (44.3% vs. 33.1%; p < 0.0001). Nour-Eldin et al. [8] similarly reported that the presence of emphysema was statistically significant for Ptx development in both Cx and NCx techniques (p < 0.001). However, some studies did not find emphysema to be a statistically significant risk factor for Ptx [9,13,17,18]. In our study “presence of emphysema” was defined specifically as emphysema along the needle tract, whereas some other studies may have simply reported the presence or absence of emphysema in general, which could partly explain these discrepancies.
Lesion size is another potential risk factor for Ptx. Targeting smaller lesions may require more needle manipulation, which could increase Ptx rates [8]. However, the literature reports conflicting results regarding this issue. In the present study, when lesions were categorised as ≤ 20 mm and > 20 mm, Ptx rates were 19.5% (29/148) and 26.2% (95/362), respectively, while some studies have reported higher Ptx rates for smaller lesions [8,9,13,19]. However, we did not observe a statistically significant relationship in our cohort (p > 0.05) (Table 2). This finding may reflect the expertise of the interventional radiologists performing the biopsies. Indeed, other studies have also reported no correlation between lesion size and Ptx [6,11,20,21]. Hiraki et al. [10], however, found that smaller lesion size was a significant risk factor for Ptx in univariate analysis, but this association did not remain significant in multivariate analysis.
Our definition of traversed aerated lung parenchyma was similar to those used in previous studies [6,7,9, 15,22,23]. Several studies have reported that increased length of aerated lung traversed, i.e., targeting deeper lesions, is associated with higher Ptx rates [6,7,9,10,13,20, 22,23]. In our cohort, patients with ≤ 20 mm, 21-39 mm, and ≥ 40 mm of traversed aerated lung parenchyma had Ptx rates of 29.82%, 19.92%, and 26.51%, respectively. These results are generally consistent with the literature, showing a tendency toward higher Ptx incidence when the traversed lung parenchyma exceeds 4 cm, although this difference was not statistically significant (p > 0.05). Regression analysis did not demonstrate a significant causal relationship. Nevertheless, similarly to Khan et al. [6] and Yeow et al. [9], we observed a trend toward higher Ptx rates when the traversed parenchymal length was ≤ 2 cm. Hiraki et al. [10] and Kuban et al. [7], in their regression analyses, reported a significant increase in Ptx risk with increasing depth of traversed aerated lung. Graffy et al. [15] noted a statistically significant increase in Ptx for lesions located at depths of 6 cm or greater. It has been suggested that deeper and smaller lesions may require more precise needle guidance, potentially causing pleural injury [10]. A key distinction of our study is that we excluded pleural-based and pleura-originating lesions. This specific lesion selection and the experience of the interventional radiologists performing the procedures may explain the differences in our findings compared to other studies.
Regarding lesion location, the literature classifies lesions based on lobes (upper, middle, lower), zones (upper, middle, lower), anatomical location (pleural, parenchymal, mediastinal), and bronchovascular tree distribution (central vs. peripheral). In our study, because we focused on traversing lung parenchyma and excluded pleural-based or pleura-originating lesions, we adopted a lobar classification for location analysis. Ptx incidence was 20.9% (60/286) in upper lobe lesions, 27.4% (57/208) in lower lobe lesions, and 43.75% (7/16) in middle lobe lesions. These differences were statistically significant (p < 0.05). Kuban et al. [7] reported Ptx incidences of 35% (775/2241) in upper lobe lesions, 24% (413/1726) in lower lobe lesions, and 33% (98/295) in middle lobe lesions. Yeow et al. [9] and Ohno et al. [20] found no association between lesion location and increased Ptx risk. Hiraki et al. [10] and Kim et al. [12] classified lesions as upper, middle, and lower lobes. Kim et al. [12] did not find a statistically significant association between location and Ptx development. Hiraki et al. [10], in univariate analysis, reported a higher Ptx risk in lower lobe lesions, and multivariate analysis identified lower lobe location as an independent risk factor for Ptx. Similarly, in our study, lower lobe lesions exhibited higher Ptx incidence compared to upper lobe lesions. Notably, middle lobe lesions showed markedly higher Ptx incidence than both upper and lower lobes. We hypothesise that the presence of minor fissures and anatomical variations in the middle lobe may contribute to this increased incidence.
Regarding approach to the lesion, we observed a lower Ptx incidence with anterior approaches compared to lateral and posterior approaches, although this difference was not statistically significant (p = 0.051). Regression analysis, however, demonstrated that the lateral approach was associated with a significantly higher Ptx risk compared to the anterior approach (p < 0.05, OR: 2.46, CI: 1.18-5.12), and the posterior approach was also associated with higher Ptx risk relative to the anterior approach (p < 0.05, OR: 2.09, CI: 1.15–3.78) (Table 4). In the literature, Hiraki et al. [10], Ohno et al. [13], and Laurent et al. [20] reported no statistically significant differences in Ptx risk based on approach. Conversely, Kuban et al. [7] found that anterior approaches were associated with increased Ptx risk compared to lateral and posterior approaches. We believe that in the lateral approach, patient positioning and stabilisation are more challenging, and similarly, the prone position required for posterior approaches may reduce patient comfort, leading to increased needle movement and greater pleural injury, which may explain the higher incidence of Ptx observed with these approaches.
In our study, the use of 18G NCx was associated with an increased incidence of Ptx compared to 19G Cx. This finding is supported by the studies of Kuban et al. [7], Nour-Eldin et al. [8], and Geraghty et al. [11]. Conversely, we observed that the use of 17G Cx significantly reduced Ptx rates compared to 18G NCx (Table 5). In their study, Nour-Eldin et al. [8] reported a lower Ptx incidence in the NCx group (23.2%, 77/332) compared to the Cx group (27%, 86/318), although this difference was not statistically significant. Malone et al. [16] reported Ptx incidences of 34% (33/96) in the 19G Cx group and 28% (40/145) in the 17G Cx group, also without statistical significance. In our study, the 18G NCx group (38.6%, 39/101) exhibited higher Ptx rates compared to the 17G Cx group, both without OBC (19.3%, 19/98) and with OBC (10.2%, 5/49). Regression analysis indicated that the use of 18G NCx was associated with increased Ptx compared to the 17G Cx system. Additionally, the 19G Cx system showed a statistically significant increase in Ptx risk relative to the 17G Cx system. We also found that the presence of T1PC was associated with a statistically significant reduction in Ptx incidence, with a meaningful correlation between T1PC presence and absence of Ptx (p < 0.001) (Table 4). De Filippo et al. [14] demonstrated that the absence of T1PC was significantly associated with Ptx development (p < 0.001). Soylu et al. [24], in their study on 18G NCx biopsies, reported a significant relationship between the absence of T1PC and Ptx development in patients without emphysema (p < 0.001), but no significant association in patients with emphysema. In our study, the 17G Cx system resulted in a significantly higher rate of T1PC formation compared to the 19G Cx and 18G NCx systems (p < 0.001) (Table 6), and we believe this statistically significant increase in T1PC acts similarly to OBC, thereby contributing to a reduction in Ptx rates.
Regarding OBC usage, we observed a statistically significant difference in Ptx incidence between patients with and without OBC. The Ptx rate was 12.1% in the OBC group versus 28.9% in the non-OBC group (p < 0.001). Multivariate regression analysis indicated that not using OBC was associated with an increased risk of Ptx compared to OBC use (p < 0.05, OR: 0.39, CI: 0.21–0.74). When analysing OBC subgroups, the Ptx incidence was 10.2% in the 17G Cx OBC group (versus 19.4% in the 17G Cx non-OBC group) and 12.2% in the 19G Cx OBC group (versus 28.6% in the 19G Cx non-OBC group) (Table 2). These results indicate that OBC use substantially reduces Ptx incidence at the subgroup level. The literature demonstrates considerable variation in reported Ptx incidences despite similar techniques. Lang et al. [25] reported a Ptx rate of 9% (4/45) in the OBC group and 47% in the non-OBC group, with a statistically significant difference in univariate analysis (p < 0.001). Graffy et al. [15], in a large patient population, reported significantly lower Ptx rates with OBC (30%, 482/145) compared to non-OBC (44%, 352/154; p < 0.001). Perl et al. [23], also in a large cohort, reported Ptx incidence of 10.7% (45/419) with OBC and 15.4% (69/449) without OBC (p < 0.05), and they found a statistically significant reduction in Ptx with OBC in the 17G Cx system, but not in the 19G Cx system. Turgut et al. [21] reported Ptx in 14.1% (13/91) of OBC patients versus 26.3% (45/171) in non-OBC patients, with statistical significance (p = 0.01). Malone et al. [16] noted a reduction in Ptx with OBC, although the difference was not statistically significant (26% vs. 35%, p = 0.12). Some smaller studies, however, found no significant difference in Ptx incidence with OBC use [26,27].
In our study, when examining the development of Ptx in relation to the number of samples obtained, univariate analysis revealed a statistically significant increase in Ptx risk when ≥ 3 samples were collected (p < 0.05). Multivariate regression analysis also demonstrated a statistically significant association (p < 0.05). Nour-Eldin et al. [8] reported a statistically significant increased Ptx risk with more than one sample in the NCx system, whereas no significant difference was observed in the Cx system. This has been attributed to the ability of the Cx system to access the lesion directly and obtain multiple samples without causing multiple pleural injuries. While we share a similar view, we believe that in the Cx system, multiple sampling around small lesions can increase parenchymal injury, thereby elevating the risk of Ptx. Yeow et al. [9], Hiraki et al. [10], and Kim et al. [12] reported no significant association between the number of samples and the development of Ptx in their studies.
Regarding the number of pleural passes, our study found a higher incidence of Ptx when ≥ 3 passes were performed compared to 1-2 passes. Univariate analysis revealed a statistically significant difference (p < 0.05), and multivariate regression analysis confirmed a significant association between ≥ 3 pleural passes and Ptx development (p < 0.05). Similar findings have been reported by Kuban et al. [6], Kim et al. [7], and Khan et al. [12]. Ohno et al. [20] demonstrated that the incidence of Ptx after the first pleural puncture was 18%, increasing significantly to 53% and 73% after the second and third pleural passes, respectively. However, some studies have found no significant relationship between the number of pleural passes and Ptx development [8,11].
Tube-requiring pneumothorax
In our study, no statistically significant difference was observed in tube-requiring Ptx between patients aged ≤ 60 and > 60 years, with rates of 6.2% (12/195) and 6.7% (21/315), respectively. Kim et al. [12] reported that patient age did not significantly affect the development of Ptx requiring catheter placement (non-catheter group: 62.6 ± 12.1 vs. catheter group: 65.1 ± 12.2, p = 0.225). Consistent with our findings, Hiraki et al. also reported that advanced age was not statistically associated with tube-requiring Ptx [10]. However, Kuban et al. [7] identified a significant relationship between advanced age and the development of tube-requiring Ptx in both univariate and multivariate analyses.
Regarding sex, the incidence of tube-requiring Ptx in our study was 7.6% (26/340) in male patients and 4.1% (7/170) in female patients. Although males had a higher incidence, the difference was not statistically significant. Similarly, Kim et al. [12] found no statistically significant difference between sexes in catheter requirement (male: 6/65; female: 30/162; p = 0.16). Hiraki et al. [10] reported tube-requiring Ptx rates of 7/158 in females and 47/294 in males. In univariate analysis, males had a significantly higher risk; however, multivariate regression showed only a weak statistical association between male sex and tube-requiring Ptx (p = 0.05) [10].
When examining Ptx rates according to the presence or absence of emphysema along the needle path, cases with emphysema had higher incidences of tube-requiring Ptx, although the difference was not statistically significant. Hiraki et al. [10] and Kuban et al. [7] reported a significant association between the presence of emphysema and tube-requiring Ptx in both univariate and multivariate analyses. Similarly, Cox et al. [19] and Kim et al. [12] reported that emphysema presence was statistically associated with the development of tube-requiring Ptx (p < 0.01).
When lesion size was categorised as ≤ 20 mm and > 20 mm, tube-requiring Ptx occurred in 6.4% (10/148) and 6.8% (23/362) of cases, respectively, with no statistically significant difference (p > 0.05). Kim et al. [12] also found no significant difference regarding catheter requirement between lesion size groups (non-catheter group: 3.1 ± 1.8 mm vs. catheter group: 3.1 ± 2.1 mm, p = 0.937). Similarly, Hiraki et al. [10] reported no significant difference in tube-requiring Ptx rates based on decreasing lesion size (p = 0.15), consistent with our findings. Kuban et al. [7], however, observed significantly higher chest tube placement in lesions ≤ 20 mm (17.8%) compared to lesions > 30 mm (10.5%).
Regarding traversed aerated lung parenchyma, tube-requiring Ptx rates were 3.5% (6/171), 7% (18/256), and 10.8% (9/185) for ≤ 20 mm, 21-39 mm, and ≥ 40 mm, respectively. Our results align with the literature, showing that the incidence increases with lesion depth, although not statistically significant (p > 0.05). Kuban et al. [7] reported corresponding rates of 4%, 16%, and 28% and found a significant relationship between lesion depth and tube-requiring Ptx in both univariate and multivariate analyses. Kim et al. [12] also reported a significant difference regarding catheter requirement between lesion depths (non-catheter: 2.2 ± 1.9 cm vs. catheter: 2.8 ± 1.7 cm, p = 0.033) and identified a significant multivariate association when the pleura-to-target distance exceeded 1.3 cm. Hiraki et al. [10] similarly found that increased lesion depth was significantly associated with tube-requiring Ptx in both univariate and multivariate analyses.
In terms of lesion location, the incidence of tube-requiring Ptx was 6.9% (20/286) in upper lobe lesions, 5.7% (12/208) in lower lobe lesions, and 6.2% (1/16) in middle lobe lesions. Consistent with the literature, a slight increase in incidence was observed in the upper and middle lobes compared to the lower lobe, although this was not statistically significant. Kim et al. [12] found no significant difference in tube-requiring Ptx according to location (p = 0.49), grouping lesions as upper-middle versus lower lobe (upper-middle: 23/701, lower: 13/526). Hiraki et al. [10] reported rates of 37/232 for upper-middle lobes and 17/220 for lower lobes. While univariate analysis showed a significant increase in tube-requiring Ptx in upper-middle lobes (p = 0.007), multivariate analysis did not confirm this association (p = 0.64). Kuban et al. [7] reported 18% in upper lobes, 17% in middle lobes, and 10% in lower lobes.
Regarding lesion approach, we observed lower tube-requiring Ptx incidence with lateral approaches compared to anterior and posterior approaches: anterior 6.6% (8/120), posterior 7.1% (22/309), and lateral 3.7% (3/81), without statistical significance (p > 0.05). Kim et al. [12] similarly reported no significant differences (p = 1), although they classified approaches as supine versus prone, rather than anterior, posterior, and lateral. Hiraki et al. [10] reported rates of 34/193 for supine and 20/256 for prone positions. While univariate analysis showed a significant difference for supine position (p = 0.002), multivariate analysis did not reveal a significant association. Kuban et al. [7] reported higher tube-requiring Ptx in anterior (20%), posterior (12%), and lateral (14%) approaches (p < 0.001), with multivariate analysis indicating anterior approach as a significant risk factor compared to lateral (p = 0.0066) and posterior (p < 0.0001) approaches. The differences between our study and Kuban et al.’s [7] may be explained by procedural variations: they did not attempt manual aspiration, and all patients were followed in the supine position during post-procedural recovery, irrespective of approach. Additionally, some previous studies have suggested that positioning the patient on the biopsy side during the post-procedural recovery period may reduce the rate of chest tube placement [28,29]. Some authors have also shown that catheter or needle aspiration alone may prevent the need for chest tube placement in a subset of these patients [30,31]. This indicates that factors such as post-procedural manual aspiration and post-procedural resting position play a role in the development of tube-requiring Ptx and should be considered in relevant studies.
In our study, the incidence of tube-requiring Ptx was 3.97% (4/101) with the 18G NCx system, 3.06% (3/98) with the 17G Cx system, 11.1% (19/171) with the 19G Cx system, 0% (0/49) with the 17-18G CxOBC system, and 7.7% (7/84) with the 19-20G CxOBC system. The differences between methods were statistically significant (p < 0.05). We observed increased incidence of tube-requiring Ptx despite smaller needle diameters in 17G Cx (with or without OBC), 18G NCx, and 19G Cx (with or without OBC). Multivariate regression analysis showed a statistically significant association between 17G Cx needle use and tube-requiring Ptx compared to 19G Cx needle use (p < 0.05, OR: 6.01, 95% CI: 1.71-21.05). No significant difference was observed between 17G Cx and 18G NCx needle use (p > 0.05). A literature review revealed that most studies have evaluated 18G and 19G needles, with less comparison involving 17G needles. Various needle sizes were used, but the relationship with tube-requiring Ptx was often not addressed, and some large studies did not consider needle size as a risk factor [7,10-12, 15,18,19,24,32]. Our study differs by separately evaluating NCx and Cx systems and OBC use as independent risk factors. Malone et al. [16] reported tube-requiring Ptx incidences of 18% with 17G and 10% with 19G needles, without statistically significant differences. Geraghty et al. [11] found no effect of 18G or 19G Cx needles on tube-requiring Ptx rates. Kuban et al. [7], in contrast, reported significantly higher tube-requiring Ptx rates with 18G needles compared to 19G in both univariate and multivariate analyses. In our study, however, 18G needles did not result in higher tube-requiring Ptx compared to 19G, and we observed a trend of decreasing Ptx incidence with larger needle diameters. We identified that 17G needle use was associated with an increased rate of T1PC compared to 18G and 19G needles. We hypothesise that this increased T1PC may function similarly to OBC, contributing to the observed reduction in tube-requiring Ptx incidence.
Regarding OBC use, tube-requiring Ptx occurred in 5% (7/140) of cases with OBC and 7% (26/370) without OBC. Although a decrease in Ptx incidence was observed with OBC, it was not statistically significant (p > 0.05). When analysing subgroups according to needle diameter, comparisons between patients with and without OBC in 17-18G Cx and 19-20G Cx systems revealed statistically significant differences (p < 0.05). In large population studies, Perl et al. [23] reported lower tube-requiring Ptx incidence in patients receiving OBC compared to those without OBC (3.1% vs. 5.8%), but this difference was not statistically significant. Bourgouin et al. [26], in a cohort of 140 patients, reported 7.7% (4/52) incidence with OBC versus 8% (8/88) without OBC, without statistical significance. Herman and Weisbrod [27], in 93 patients, also found no significant difference with OBC use. Malone et al. [16] reported a significant reduction with OBC (9% vs. 18%, p = 0.048). Graffy et al. [15], in patients with intraparenchymal blood patch, demonstrated a significantly lower rate of tube-requiring Ptx (3.1% vs. 6.8%, p < 0.001). We believe that technical variations, such as the use of blood or patch techniques, may account for these differences in outcomes. More standardised and homogeneous techniques in large population studies could clarify the relationship between OBC and tube-requiring Ptx more definitively.
Regarding the number of samples obtained, in our study, univariate analysis revealed a significantly higher risk of tube-requiring Ptx in cases with ≥ 3 samples (14/91, 15.4%) compared to those with ≤ 2 samples (19/419, 4.5%) (p < 0.05). Multivariate regression analysis also demonstrated a statistically significant association (p < 0.001, OR: 4.26, 95% CI: 1.82-9.94). In the study by Kim et al. [12], tube-requiring Ptx occurred in 7/344 patients with ≤ 2 samples and in 29/883 patients with ≥ 3 samples, without statistically significant difference (p = 0.34). Hiraki et al. [10] also reported no significant association between sample number and the development of tube-requiring Ptx. We believe that the main reason our results differ from the literature is that one of our patient groups underwent biopsies using the 18G NCx system. In NCx systems, each sample also results in a new pleural puncture.
Regarding the number of pleural passes, univariate analysis in our study showed a significantly increased risk of tube-requiring Ptx with ≥ 3 pleural passes (4/6, 66.7%) compared to ≤ 2 passes (29/504, 5.8%) (p < 0.001). Multivariate regression analysis also revealed a statistically significant association (p < 0.05, OR: 28.84, 95% CI: 2.80-296.11). Kuban et al. [7] and Kim et al. [12] reported that an increased number of pleural passes was associated with higher tube-requiring Ptx incidence, and this increase reached statistical significance.
Type 1 parenchymal contusion
In literature reviews, the risk of contusion following CTTB has been reported to range from 2.9% to 54% [3]. However, we agree with Tai et al.’s opinion [17] that this complication is underreported. In fact, every percutaneous thoracic biopsy is associated with varying degrees of pulmonary haemorrhage along the needle tract or in the perilesional lung tissue, manifested as focal increased attenuation [6,9]. In a study of 872 patients, Soylu et al. [24] reported a T1PC incidence of 33.7% with the 18G NCx system. In our study, T1PC was more common (57.8%). We evaluated the relationship between needle tract contusion and factors including age, sex, emphysema, lesion size, location, lesion approach, method (needle gauge), length of aerated lung traversed, number of samples, and number of pleural passes.
In our study, T1PC incidence did not differ significantly between patients aged ≤ 60 years (80/133, 60.2%) and > 60 years (134/237, 56.5%). Soylu et al. [24] reported slightly higher T1PC incidence in patients ≤ 60 years compared to those > 60 years (38% vs. 30%), but age was not statistically significant in relation to needle tract contusion. Similarly, Yeow et al. [9,18], Nour-Eldin et al. [33], and Wattanasatesiri et al. [34] reported no significant effect of advanced age on T1PC rates.
Regarding sex, T1PC incidence was 55.9% in men and 36.7% in women. Although higher in men, the difference was not statistically significant. Soylu et al. [24] similarly reported slightly higher T1PC incidence in men compared to women (34% vs. 31%), without statistical significance.
When evaluating the presence or absence of emphysema along the needle tract, T1PC incidence was 58.3% in patients with emphysema and 56.1% in those without, with no statistically significant difference. Soylu et al. [24] reported similar findings, showing no significant difference in T1PC rates based on emphysema presence. Yeow et al. [18], Nour-Eldin et al. [33], and Wattanasatesiri et al. [34] also found no significant association between emphysema and T1PC incidence.
Regarding lesion size, T1PC incidence was 60.2% in lesions ≤ 20 mm and 57% in lesions > 20 mm. Although smaller lesions showed a non-significant increase in T1PC, the trend was apparent. Topal et al. [35] reported a similar trend, with a non-significant increase in T1PC risk for smaller lesions. Yeow et al. [9] and Laurent et al. [13] reported a significantly higher T1PC risk in small lesions. Yeow et al. [18], Khan et al. [6], and Nour-Eldin et al. [33] also reported higher T1PC rates in small lesions, sometimes reaching statistical significance. We believe that these discrepancies in the literature are attributable to differences in technique, operator experience, and needle gauge, leading to heterogeneity and variable results across studies.
In the current study, T1PC incidence was 55.3% (78/141) in lesions with ≤ 20 mm of aerated lung traversed, 58.2% (107/184) in lesions with 21-39 mm, and 64.4% (29/45) in lesions with ≥ 40 mm, with this increase not reaching statistical significance. In the study by Soylu et al. [24], lesions with depth > 3 cm had a 40% incidence of T1PC, compared to 24% in lesions 0.5-3 cm deep, and they reported a statistically significant increase in T1PC with increasing lesion depth. Similarly, Yeow et al. [9] reported a statistically significant rise in T1PC incidence with increasing lesion depth. They found a 10-fold higher T1PC risk in lesions with a parenchymal depth ≥ 2.1 cm compared to pleura-adjacent lesions. In our study, we excluded lesions that did not traverse aerated lung parenchyma. As expected, these pleura-based lesions carry a lower risk of complications compared to biopsies passing through aerated lung parenchyma. We believe that this exclusion criterion accounts for the differences in our findings compared to previous literature. This methodological distinction is one feature that differentiates our study from other published reports. Yeow et al. [18], Khan et al. [6], Topal and Berkmanb [35], and Nour-Eldin et al. [33] have all reported a statistically significant increase in T1PC incidence with increasing lesion depth. It appears that in deeper lesions, the needle is more likely to traverse vascular structures in the parenchyma, thereby increasing the risk of parenchymal contusion.
Regarding lesion location, T1PC incidence was 57.4% (113/197) in upper lobe lesions, 59.7% (95/164) in lower lobe lesions, and 42.9% (6/14) in middle lobe lesions, with no statistically significant differences observed. Yeow et al. [9], Soylu et al. [24], and Wattanasatesiri et al. [34] similarly reported no significant association between lesion location and T1PC. In contrast, Nour-Eldin et al. [33] reported a statistically significant increase in T1PC risk for lesions in the lower and middle zones compared to the upper zone; however, their study used a zonal classification, differing from our methodological approach.
Regarding lesion approach, T1PC incidence in our study was lower with the lateral approach (47.5%) compared to the anterior (61.6%) and posterior (59.2%) approaches, although this difference was not statistically significant. Similarly, Soylu et al. [24] reported no statistically significant association between lesion approach and T1PC incidence.
Significant differences were observed between needle gauge and T1PC incidence. T1PC rates were 76.5% (75/98) with the 17G Cx system, 48.5% (49/101) with the 18G NCx system, and 52.6% (81/171) with the 19G Cx needle. Multivariate logistic regression analysis showed that the use of a 17G needle was significantly associated with increased T1PC risk compared to 18G NCx and 19G Cx needles (p < 0.001) (Table 6). Topala and Berkmanb [35] demonstrated that using 18G, 20G, or 22G needles did not result in a statistically significant increase in T1PC risk. Yeow et al. [9] used needles ranging from 16G to 20G and reported no significant association between needle gauge and T1PC in small lesions. In the present study, procedures were performed entirely based on operator preference without adjusting for smaller lesion size or needle gauge. We observed that the relationship between 17G needle use and T1PC has not been sufficiently investigated in the literature.
Regarding the number of samples, in univariate analysis, T1PC incidence was 55.1% (167/303) in patients with ≥ 3 samples and 70.1% (47/67) in patients with ≤ 2 samples. Statistically, T1PC was significantly more common in the ≥ 3 samples group (p < 0.05). However, multivariate analysis did not reveal a statistically significant association. There is limited literature on this topic. We hypothesise that an increased number of samples may lead to longer procedure times and more needle manoeuvres, thereby increasing parenchymal injury.
Regarding the number of pleural passes, T1PC incidence was higher with ≥ 3 passes (75%) compared to ≤ 2 passes (57.7%), although this was not statistically significant. Khan et al. [6] reported a higher risk of T1PC with 2 pleural passes (33%) compared to 3-4 passes (21%) and 1 pass (0%). Tai et al. [17] found that the number of pleural passes did not significantly affect T1PC incidence. In both our study and the literature, pleural pass number has been investigated; however, differences in biopsy technique and lesion location can affect interpretation. For example, with a Cx system, a single biopsy may traverse two lobes, resulting in three pleural passes, whereas with an NCx system, obtaining three samples from a single lesion also results in three pleural passes. These examples differ in technique and complication risk. Therefore, large-scale studies using standardised methodology are needed to more accurately assess this factor.
Haemoptysis, haemothorax, air embolism, and mortality
In our study, haemoptysis was observed in 14% of all patients. The reported incidence of haemoptysis in the literature ranges between 0.5% and 14.4% [3]. Our findings are consistent with the literature. Haemothorax, a rare complication reported in very few studies, occurred in one patient in our cohort [36,37]. No cases of air embolism or death were observed in our study.
The main limitations of our study are related to its retrospective design. First, the study was limited to patients pre-selected for biopsy. Second, the assessment of emphysema was performed subjectively, which could lead to interobserver variability. Future studies should consider quantitative assessment of emphysema to obtain more objective results. Third, operator experience was not evaluated in our study. Fourth, the choice of technique and needle gauge was left to the discretion of the operator. Fifth, procedure durations were not assessed. Despite these limitations, our results reflect data obtained using state-of-the-art equipment and a range of techniques in clinical practice, as well as our institutional experience.
Conclusions
In CT-guided transthoracic lung biopsies, the incidence of Ptx and chest tube insertion is influenced by technical parameters, needle size, pleural puncture count, and the number of specimens obtained. In our cohort, the use of OBC and 17G coaxial systems was associated with lower Ptx rates. Conversely, lateral or posterior approaches, the presence of emphysema along the needle tract, ≥ 3 pleural punctures, and ≥ 3 specimens significantly increased Ptx risk. These findings highlight the importance of tailoring the biopsy technique to both patient and lesion characteristics to minimise complications.