Introduction
Prostate cancer (PCa) is the second most frequently diagnosed malignancy and a leading cause of cancer-related mortality among men worldwide, with over 1.4 million new cases and 375,000 deaths annually [1]. The global burden of PCa continues to rise, largely due to aging populations and increased prostate-specific antigen (PSA)-based screening. Accurate diagnosis and risk stratification are essential to guide treatment decisions, avoid overtreatment of indolent tumors, and identify clinically significant disease early.
Limitations of standard transrectal ultrasound-guided biopsy
For decades, the diagnostic cornerstone of PCa has been systematic transrectal ultrasound-guided biopsy (TRUS-GB). This approach involves obtaining 10-12 random cores, typically from the peripheral zone, but suffers from suboptimal sensitivity and random sampling errors [2,3]. Major limitations include: missed clinically significant PCa (csPCa), especially in anterior or apical regions, overdiagnosis of clinically insignificant tumors (Gleason 3 + 3),variable detection rates depending on operator experience, risk of infection, bleeding, and sepsis [4]. Consequently, there has been a paradigm shift toward image-guided targeted biopsy techniques to improve diagnostic yield and reduce unnecessary sampling.
Role of multiparametric MRI and Prostate Imaging Reporting and Data System
Multiparametric MRI (mpMRI) of the prostate, integrating T2-weighted imaging (T2WI), diffusion-weighted imaging (DWI), and dynamic contrast-enhanced (DCE) sequences, has revolutionized PCa diagnostics [5–7]. The introduction of the Prostate Imaging Reporting and Data System (PI-RADS), now updated to version 2.1, standardized lesion evaluation and reporting, improving reproducibility and clinical decision-making [8].
Key advantages of mpMRI
mpMRI provides accurate localization and detailed characterization of suspicious lesions, enabling precise identification of areas that may harbor malignancy. It allows differentiation between csPCa and insignificant PCa, which is essential to reduce overtreatment of indolent disease.
mpMRI improves the detection of tumors often missed by conventional TRUS-GB, particularly those situated in the anterior and transition zones of the prostate.Furthermore, mpMRI serves as a critical tool for guiding targeted biopsies and supporting active surveillance protocols, facilitating precise risk stratification and optimized patient management [9].
MRI-targeted biopsy approaches
MRI has enabled three principal strategies for targeted biopsy [10–12]:
cognitive registration biopsy – performed using prior MRI knowledge during real-time TRUS;
MRI/ultrasound (US) fusion-guided biopsy – fuses MRI lesions with live US images;
In-bore MRI-guided biopsy (IB-MRGB) – performed entirely under real-time MRI guidance.
Among these, IB-MRGB is considered the most precise technique, enabling direct lesion targeting within the MRI bore. It is particularly effective for small, anterior, or apical tumors, often undetectable with TRUS [13,14]. Several meta-analyses confirm that IB-MRGB demonstrates superior per-core diagnostic yield, minimizes unnecessary sampling, and reduces overdiagnosis of indolent tumors [15–17].
Aim and scope of this review
Despite its growing clinical relevance, IB-MRGB remains underutilized due to cost, logistical demands, and limited access to MRI time. Nevertheless, recent advances in robotic assistance, faster imaging protocols, and software-guided targeting are improving feasibility and efficiency.
This review provides a comprehensive summary of current evidence, technical considerations, and future directions for IB-MRGB, emphasizing its role in precision diagnostics, treatment planning, and active surveillance.
Technical overview of IB-MRGB
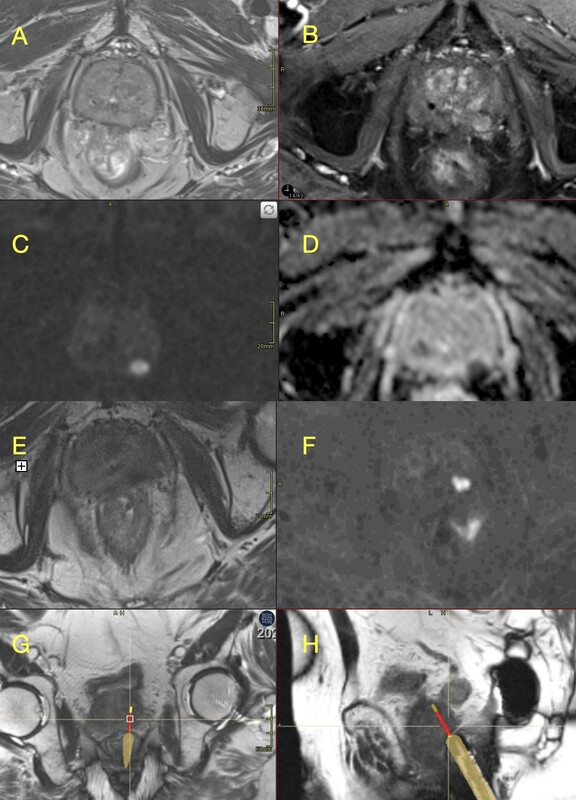
IB-MRGB is performed on 1.5T or 3.0T MRI scanners equipped with a dedicated interventional platform. Patients are typically positioned prone to facilitate rectal access and align the biopsy device (Figure 1). The standard workflow includes:
planning mpMRI with T2WI, DWI, and optional DCE sequences for lesion localization according to PI-RADS v2.1;
software-assisted trajectory planning for each target lesion;
stepwise or real-time needle insertion using MRI-compatible 18G needles;
verification with rapid T2WI before tissue sampling.
Figure 1
Patient positioning for in-bore magnetic resonance image (MRI)-guided prostate biopsy. The patient is placed in the prone position with a dedicated positioning device and coil system, allowing transrectal access and stable probe guidance within the MRI bore
Platforms such as Soteria RCM 1.0 and DynaTRIM enhance reproducibility and accuracy, with 2-4 targeted cores per lesion typically sufficient for diagnosis [8,14].
Diagnostic accuracy
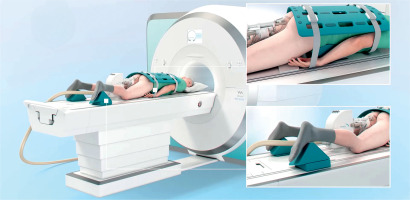
IB-MRGB provides the highest sensitivity (80-96%) and specificity (82-90%) for csPCa among MRI-targeted techniques [9,11,12,16]. Meta-analyses confirm that IB-MRGB achieves superior per-core diagnostic yield and reduces the number of biopsy cores needed. It is especially valuable in patients with prior negative TRUS-GB and in detecting anterior or apical tumors. For subcentimeter lesions, approximately 5 mm in diameter, integrating DWI with apparent diffusion coefficient (ADC) mapping facilitates biopsy guidance by directing the needle toward the segment with the lowest ADC values, which is presumed to represent the most aggressive tumor focus [16] (Figure 2).
Figure 2
Example of Prostate Imaging for Recurrence Reporting (PI-RR) 4 lesion on multiparametric magnetic resonance imaging (mpMRI) with in-bore targeting (Gleason 4 + 3). A) Axial T2-weighted image showing a low-signal focal lesion in the left peripheral zone (mid-gland). B) Dynamic contrast-enhanced (DCE) imaging demonstrates early enhancement. C, D) Diffusion-weighted imaging (DWI) and corresponding apparent diffusion coefficient (ADC) map reveal diffusion restriction. E-H) In-bore MRI-guided biopsy: T2-weighted (E) and DWI (F) images for targeting; real-time T2-weighted axial (G) and sagittal (H) images during tissue sampling
Clinical applications
IB-MRGB has multiple clinical applications across the spectrum of PCa management.
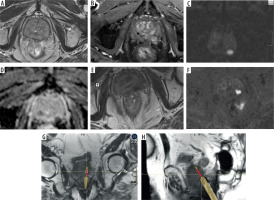
First, IB-MRGB is highly valuable in biopsy-naïve patients. By enabling direct, MRI-targeted sampling of suspicious lesions, the technique significantly improves the detection of csPCa while simultaneously reducing the overdiagnosis of indolent Gleason 3 + 3 tumors [12,16]. This approach aligns with the principles of precision medicine by focusing on clinically meaningful disease. Second, IB-MRGB is particularly beneficial for patients with prior negative TRUS-GB but persistent clinical suspicion. In this setting, IB-MRGB demonstrates a high diagnostic yield, often identifying anterior or apical tumors that are frequently missed with systematic TRUS biopsies [10,11] (Figure 3). As a result, it provides a crucial diagnostic tool for patients with rising PSA levels or ongoing suspicion of PCa despite prior negative results. Third, IB-MRGB plays an increasingly important role in active surveillance protocols.
Figure 3
Example of Prostate Imaging Reporting and Data System (PI-RADS) 4 lesion on multiparametric magnetic resonance imaging (mpMRI) with in-bore targeting (Gleason 3 + 4). A) Axial T2-weighted image showing a mildly hypointense focal lesion in the left transitional zone (mid-gland). B, D) Diffusion-weighted imaging (DWI) and the corresponding apparent diffusion coefficient (ADC) map demonstrate diffusion restriction. C) Dynamic contrast-enhanced imaging shows subtle early enhancement of the lesion. E-H) In-bore MRI-guided biopsy: T2-weighted (E) and DWI (F) images for lesion targeting; real-time T2-weighted axial (G) and DWI (H) images acquired during tissue sampling
It provides reliable confirmatory biopsies at the time of initial risk stratification and can monitor disease progression over time with high accuracy. By focusing sampling on MRI-visible lesions, IB-MRGB reduces the need for repeated systematic biopsies, thereby minimizing patient morbidity while maintaining oncologic safety [18].
Finally, IB-MRGB is an essential tool in planning and guiding focal therapy.
By offering precise lesion localization, it facilitates accurate mapping for minimally invasive treatments such as high-intensity focused ultrasound (HIFU), cryotherapy, or focal laser ablation [13,19,20]. This integration supports the transition toward personalized, lesion-targeted therapies in PCa management. Collectively, these applications highlight IB-MRGB as a versatile and high-value technique that enhances diagnostic precision, informs treatment planning, and supports modern approaches to active surveillance and focal therapy.
Discussion
IB-MRGB has emerged as the most precise MRI-targeted biopsy method for diagnosing PCa. Unlike cognitive or fusion biopsy, IB-MRGB offers direct real-time targeting of suspicious lesions with the needle trajectory fully visualized on high-resolution mpMRI [12,16]. This is particularly valuable for small, anterior, apical, or transition zone lesions that are frequently under-sampled by standard TRUS-GB [3].
Meta-analyses and multicenter studies consistently demonstrate the high diagnostic performance of IB-MRGB, with sensitivity for csPCa ranging from 80 to 96% and specificity from 82 to 90% [7,11,15,16]. Schoots et al. [12] confirmed superior per-core detection efficiency compared to MRI/US fusion and cognitive techniques, reducing the number of unnecessary samples (Table 1).
Table 1
Summary of recent meta-analyses evaluating in-bore magnetic resonance imaging-guided prostate biopsy (IB-MRGB) performance
| Author (year) | csPCa detection rate | Sensitivity/specificity | Key findings |
|---|---|---|---|
| Wegelin et al., 2017 [11] | Variable across studies (no single pooled estimate for IB-MRGB) | Not reported as pooled sensitivity/specificity for IB-MRGB | In-bore MRI-guided biopsy shows a higher per-core cancer detection yield than fusion and cognitive targeting, with no clear superiority of any targeting technique in overall csPCa detection. |
| Bass et al., 2022 [16] | MRI-targeted biopsy pooled csPCa detection ~0.83 | Not reported as pooled sensitivity/specificity (analysis based on detection rates) | MRI-targeted biopsy improves csPCa detection compared with TRUS-guided biopsy; no significant differences in csPCa detection were observed between in-bore, fusion, and cognitive targeting techniques. |
| Penzkofer et al., 2015 [14] | 56.7% overall cancer detection (with subgroup variation depending on clinical context); clinically relevant disease often successfully sampled | Not reported as classical sensitivity/specificity | In-bore 3T MRI-guided biopsy is clinically feasible and provides high targeting accuracy for MRI-visible lesions, with good lesion localisation compared with histopathology. |
Role in clinical practice
IB-MRGB is indicated in several key clinical scenarios:
Biopsy-naïve patients – improves csPCa detection and reduces overdiagnosis of Gleason 3 + 3 disease [12,16].
Patients with prior negative TRUS-GB – demonstrates high yield in men with persistent clinical suspicion and PSA elevation, often detecting anterior or apical tumors missed previously [10,11].
Active surveillance – plays a central role in confirmatory biopsies and longitudinal monitoring, reducing the need for systematic rebiopsy. Stavrinides et al. [18]emphasized that IB-MRGB improves detection of pathological progression while minimizing overtreatment in active surveillance protocols.
Focal therapy planning – IB-MRGB provides precise lesion localization, enabling targeted ablative procedures such as HIFU, cryotherapy, or focal laser ablation. Cornud et al. [19] reported that IB-MRGB-guided targeting supports more reliable focal therapy mapping. Elkhoury et al. [20] highlighted its role in patient selection and follow-up for focal therapies.
After radiotherapy and prior to initiating salvage treatment [21–23].
Advantages and current limitations
IB-MRGB provides several notable advantages. First, it offers the highest targeting accuracy among all MRI-guided biopsy techniques, allowing precise sampling of suspicious lesions. Second, it reduces the overall sampling burden, as fewer biopsy cores are required to achieve a reliable diagnosis. Additionally, IB-MRGB is associated with a lower risk of overdiagnosing clinically insignificant tumors, such as Gleason 3 + 3, which aligns with the principles of precision medicine. However, the technique also has important limitatins.
IB-MRGB is resource-intensive, requiring access to an MRI scanner and a specialized interventional platform, which limits its availability. The procedure is longer and more expensive compared to MRI/US fusion-guided or standard TRUS-GB.
Furthermore, small MRI-invisible csPCas (approximately 10-15%) can still be missed [17].
Despite these challenges, ongoing technological progress and better workflow integration are steadily improving the feasibility of IB-MRGB and may facilitate broader clinical adoption [12–14].
The main limitations of inbore MRIguided prostate biopsy include large prostate volume due to benign prostatic hyperplasia, lesions located in the posterior base of the gland, and specific anatomical features of the rectum, such as sharp angulation at the anorectal junction, which can hinder robotic device maneuverability and access to the target. Successful navigation in such cases requires substantial radiologist experience, careful insertion of the MRIcompatible guide, and sometimes the use of additional cushions under the patient’s hips to facilitate robot movement.
Another technical challenge arises from the different patient positioning during diagnostic MRI (supine) compared to inbore biopsy (prone), which can reduce the signaltonoise ratio and impair lesion visibility, particularly in obese patients.
The procedure is typically performed under local anesthesia, which is usually sufficient, although some patients may require analgosedation. However, prone positioning makes patient monitoring more challenging for the anesthesia team, especially since the patient remains inside the narrow MRI gantry during the procedure.
Future perspectives
The future development of IB-MRGB is closely linked to advances in imaging technology, artificial intelligence (AI), and minimally invasive interventions. Radiomics and AI are expected to play a key role in automating lesion detection and improving risk stratification, which could enhance both diagnostic accuracy and procedural efficiency [24].
Another important direction is the standardization of PI-RADS v2.1-based workflows, which will ensure reproducibility of imaging interpretation and facilitate multi-center implementation [25].
Moreover, IB-MRGB is likely to become increasingly integrated with focal therapies, enabling truly imageguided precision treatments such as HIFU, cryotherapy, and focal laser ablation [19,20].
Finally, IB-MRGB is anticipated to have an expanded role in active surveillance, as it allows for minimally invasive, high-yield follow-up biopsies that can improve disease monitoring while reducing unnecessary interventions [18].
Collectively, these developments are expected to optimize diagnostic efficiency, minimize unnecessary biopsies, and facilitate broader clinical adoption of IB-MRGB in the context of precision oncology.
Another promising direction for the development of IB-MRGB is integration with molecular imaging. In particular, the combination of prostate-specific membrane antigen (PSMA)-targeted positron emission tomography (PET) with MRI (PET/MRI) or computed tomography (PET/CT) offers highly specific detection of PCa lesions. Fusion of PSMA-PET data with mpMRI during in-bore targeting may allow for even more accurate identification of clinically significant lesions, especially in cases with equivocal MRI findings or previously negative biopsy. Early pilot experiences and conceptual frameworks suggest that PET/MRI- or PET/CT-informed in-bore biopsy may represent a new frontier in precision diagnostics [26,27].
Conclusions
IB-MRGB provides unparalleled accuracy for detecting csPCa. Its role spans biopsy-naïve cases, repeat biopsies, active surveillance, and focal therapy planning. While its use is currently limited by cost and logistical factors, integration with AI, radiomics, and minimally invasive therapies is expected to expand its clinical adoption and reinforce its value in precision medicine.