Introduction
Parkinson’s disease (PD) is the second most common neurodegenerative disorder, characterized by progressive degeneration of dopaminergic neurons in the substantia nigra (SN) and a wide spectrum of motor and non-motor symptoms. Until recently, PD diagnosis relied primarily on clinical evaluation and the exclusion of alternative etiologies. However, a major shift has occurred in the conceptual framework of the disease, with the emergence of a new biological definition of PD, grounded in the presence of pathological α-synuclein aggregates in the central nervous system (CNS).
In 2024, the International Parkinson and Movement Disorder Society formally introduced a distinction between biological and clinical definitions of PD. The term biological Parkinson’s disease refers to individuals who exhibit CNS α-synuclein pathology – confirmed through biomarkers – regardless of clinical symptomatology [1]. In parallel, a comprehensive staging model for α-synucleinopathy was proposed in The Lancet Neurology, encompassing preclinical, prodromal, and symptomatic stages, each defined by the presence of molecular biomarkers [2].
This evolving framework opens a path toward earlier detection of PD – prior to the appearance of classic motor symptoms. Within this context, magnetic resonance imaging (MRI) has gained increasing attention as a non-invasive modality capable of detecting both structural and functional changes that may accompany early α-synuclein pathology.
The potential of MRI has expanded considerably with the development of advanced techniques, including neuromelanin-sensitive MRI (NM-MRI), susceptibility-weighted imaging (SWI), quantitative susceptibility mapping (QSM), diffusion tensor imaging (DTI), functional MRI (fMRI), and ultra-high-field 7T MRI. A study published in NPJ Parkinson’s Disease demonstrated that 7T MRI enables high-resolution mapping of nigrosomes N1 and N2, as well as quantitative assessment of neuromelanin and iron accumulation – parameters that correlate strongly with motor symptom severity [3].
Furthermore, a 2021 publication in Radiology highlighted the diagnostic utility of neuromelanin and SWI imaging for distinguishing PD from atypical parkinsonian syndromes such as progressive supranuclear palsy (PSP) and multiple system atrophy (MSA) [4].
Concurrently, expert reviews underscore the need for integrating imaging biomarkers with clinical classification frameworks. The SynNeurGe project, aimed at operationalizing the biological definition of PD in research and clinical settings, recognizes MRI as a key modality for correlating clinical symptoms with underlying neurodegenerative processes [5].
In light of these developments, there is a growing need to synthesize the current knowledge regarding the diagnostic potential of MRI – not only for disease differentiation but also as a tool for identifying patients in the biological phase of PD prior to clinical manifestation [6].
The aim of this review is to provide a comprehensive overview of the current applications of MRI in the diagnosis and monitoring of PD – from conventional sequences to advanced high-resolution techniques and radiomics –and to place these tools within the emerging biological framework of PD. This article is intended as a practical guide for radiologists seeking to understand and implement MRI-based biomarkers in the context of neurodegenerative disorders.
Pathophysiological background of PDin the context of MRI
The pathophysiology of PD is rooted in neurodegenerative processes, primarily involving the progressive loss of dopaminergic neurons in the SN pars compacta (SNpc), the accumulation of pathological α-synuclein, and subsequent degeneration of the striatum and fronto-basal circuits. These changes begin long before the clinical onset of motor symptoms and represent key targets for MRI-based diagnostics.
The principal structural hallmark of PD is the gradual atrophy of the SNpc, which has been shown to correlate with the severity of motor symptoms. This atrophy particularly affects the nigrosomes – small clusters of neuromelanin-rich neurons that are highly susceptible to oxidative stress. Post-mortem studies have demonstrated that nigrosome 1 (N1) undergoes degeneration early in the disease course [7].
MRI enables indirect visualization of these processes. Iron-sensitive sequences such as SWI and QSM facilitate the assessment of pathological iron deposition in the SNpc – one of the key contributors to neurotoxicity in PD [8].
Concurrently, NM-MRI, based on T1-weighted (T1W) sequences with background signal suppression, allows for the evaluation of the volume and integrity of pigment-containing neurons in the SNpc. A reduced neuromelanin signal in this region has been shown to correlate with dopaminergic neuronal loss and greater clinical severity [9].
With the introduction of 7T MRI, it is now possible to precisely visualize nigrosomes N1 and N2 as regions of hypointensity on SWI/QSM. In healthy individuals, these regions exhibit a characteristic “swallow tail” appearance, the absence of which is considered a highly specific imaging marker for PD [10].
Degenerative changes in PD also affect the striatum, including the caudate nucleus and putamen. Although MRI cannot directly measure dopamine levels, secondary changes in volume, microstructure, and connectivity of these regions can be detected. DTI studies have shown a significant reduction in fractional anisotropy (FA) and increased radial diffusivity (RD) in white matter tracts connecting the SN with the caudate and prefrontal cortex in PD patients [11].
As PD progresses, it also affects functional brain networks. fMRI studies have demonstrated reduced connectivity within the default mode network, executive network, and cortico-basal circuits, which contributes to both motor and cognitive symptoms [12].
An increasing body of evidence suggests that neurodegeneration in PD is not limited to the nigrostriatal system but has a global distribution, affecting the limbic system, hippocampus, brainstem, and amygdala – particularly in patients with prominent non-motor symptoms such as depression, REM sleep behavior disorder (RBD), or cognitive impairment [13].
From the standpoint of MRI diagnostics, these neurodegenerative processes represent potential imaging biomarkers for both diagnosis and disease monitoring. Notably, many of these changes can be detected even before the onset of overt clinical symptoms, aligning with the emerging biological definition of PD.
Conventional MRI sequences in the diagnosis of PD
In routine clinical practice, MRI in PD is most frequently performed to exclude other structural causes of parkinsonism, such as cerebrovascular lesions, tumors, normal-pressure hydrocephalus, or corticobasal atrophy. Conventional MRI sequences – T1W, T2-weighted (T2W), and fluid-attenuated inversion recovery (FLAIR) – typically do not reveal specific changes diagnostic for PD, but may provide valuable information in the differential diagnosis of atypical parkinsonian syndromes.
Technological advances, particularly the implementation of iron-sensitive sequences such as SWI and QSM, as well as NM-MRI, have significantly improved the visualization of structural changes in the SN. However, the diagnostic sensitivity and specificity of these techniques vary substantially depending on the field strength of the MRI scanner used.
In 1.5T systems, the ability to detect subtle changes in the SN is limited. Neuromelanin imaging suffers from low contrast, and the “swallow tail” sign – typically visible in healthy controls – is usually absent, which complicates the evaluation of nigrosomal integrity. The diagnostic sensitivity of 1.5T MRI for detecting PD generally does not exceed 70% [9].
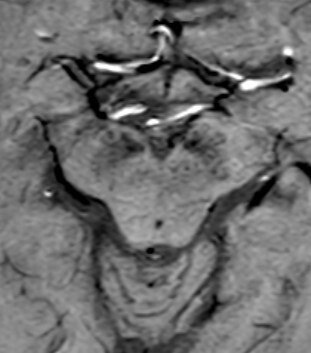
3T MRI offers significantly better contrast for neuromelanin imaging, along with moderate sensitivity for detecting iron deposition. The “swallow tail” sign is typically present in healthy individuals but disappears in patients with PD, making it a simple and practical marker in everyday clinical use [14] (Figure 1).
The highest diagnostic potential is offered by 7T MRI, which provides submillimeter spatial resolution (~0.5 mm) that enables precise delineation of nigrosomal substructures and confident identification of their presence or absence. Studies using 7T MRI have reported diagnostic sensitivities of 85-95% and specificities ranging from 85% to 92% in distinguishing PD from controls [15].
The role of radiologist experience
The interpretation of MRI in the context of PD requires specialized expertise. Several studies have demonstrated that the accurate identification of the “swallow tail” sign and assessment of neuromelanin signal depend significantly on the radiologist’s level of training. In an international study, diagnostic accuracy among neuroradiologists was markedly higher – by up to 15-20 percentage points – compared to general radiologists [15].
Therefore, it is strongly recommended that MRI interpretation in suspected PD be performed by radiologists with experience in neurodegenerative imaging, ideally with access to reference standards and curated image libraries.
Advanced MRI techniques
Technological advances in MRI have enabled the development of specialized imaging methods that go well beyond standard T1-, T2-, and FLAIR-based protocols. In the context of PD, key emerging techniques include NM-MRI, iron-sensitive methods (SWI, QSM), DTI, fMRI, and ultra-high-field imaging using 7T MRI scanners (Table 1 [15–17]).
Table 1
Comparison of magnetic resonance imaging (MRI) techniques in Parkinson’s disease (PD) diagnosis
| MRI field strength | Neuromelanin contrast | SWI/QSM quality | Ability to visualize N1 | Spatial resolution (mm) | Diagnostic sensitivity for PD (%) | Diagnostic specificity for PD(%) | Impact of radiologist experience | Primary references |
|---|---|---|---|---|---|---|---|---|
| 1.5T | Low | Limited | Rarely visible | ~1.5 | ~65-70 | ~70 | High variability; lower reliability | Schwarz et al., 2018 [16] |
| 3T | Moderate | Adequate | Usually visible | ~1.0 | ~80-85 | ~85 | Moderate; improves with training | Noh et al., 2015 [17] |
| 7T | High | Excellent | Clearly visible | ~0.5 | ~85-95 | ~85-92 | Significant impact; requires expertise | Wang et al., 2025 [15] |
NM-MRI
Neuromelanin is a pigmented polymer found in dopaminergic neurons of the SN, the concentration of which decreases with advancing neurodegeneration. NM-MRI relies on T1W sequences with background suppression (e.g., magnetization transfer contrast), allowing clear visualization of the SNpc. Several studies have demonstrated that both the volume and signal intensity of neuromelanin in SNpc are reduced even during the prodromal stage of PD [9]. The reported sensitivity of NM-MRI in detecting PD reaches up to 85%, with specificity around 88%, especially when performed on 3T or higher field-strength systems [18].
Iron-sensitive sequences: SWI and QSM
PD is associated with abnormal iron accumulation in the SN, which contributes to oxidative stress and neuronal death. SWI is sensitive to local magnetic field inhomogeneities caused by metal deposition, whereas QSM allows for precise quantification of iron distribution. Increased magnetic susceptibility in the SNpc and basal ganglia has been consistently observed in patients with PD [8].
These techniques are particularly effective at 3T and 7T field strengths, where their high resolution enables differentiation between PD and atypical parkinsonian syndromes such as PSP or MSA [8].
Structural and DTI
DTI provides microstructural information about white matter integrity. In PD, decreased FA and increased RD have been identified in nigrostriatal pathways as well as in the prefrontal cortex and limbic structures [11].
Although DTI metrics vary depending on imaging protocol, meta-analyses support their diagnostic utility – particularly when used in connectome analysis or in conjunction with functional data.
fMRI
fMRI enables the assessment of brain connectivity by measuring blood oxygen level-dependent signal changes. In PD patients, reduced functional connectivity has been documented within the default mode network, executive control networks, and cortico-basal ganglia loops [19].
These alterations are associated with both motor and cognitive symptoms, including psychomotor slowing and dementia, and may appear even in the early disease stages. fMRI is also useful in monitoring therapeutic response (e.g., to deep brain stimulation or dopaminergic therapy) and in assessing disease progression.
7T MRI – ultra-high-field imaging
7T MRI systems offer unprecedented spatial resolution (~0.5 mm), enabling detailed imaging of the SN, including nigrosomes N1 and N2. In the study by Madelung et al. [3], 7T MRI successfully distinguished healthy individuals from PD patients based on the presence or absence of N1, achieving high diagnostic accuracy – area under the curve (AUC) > 0.90.
Although the availability of 7T MRI is still limited, its application in imaging molechular biomarkers and PD-related neuroanatomy may revolutionize early diagnosis and patient classification in the near future.
The prodromal phase of PD refers to the period preceding the onset of classical motor symptoms, during which neurodegenerative changes – such as α-synuclein aggregation and dopaminergic neuronal loss – are already underway. The newly proposed biological definition of PD emphasizes that such changes can be detected via appropriate biomarkers before the onset of clinical manifestations [1,2].
MRI may play a critical role in identifying individuals in this stage, particularly those with recognized risk factors such as RBD, anosmia, depression, or LRRK2 and GBA1 mutations.
MRI and the prodromal phase of PD
The prodromal phase of PD refers to the period preceding the onset of classical motor symptoms, during which neurodegenerative changes – such as α-synuclein aggregation and dopaminergic neuronal loss – are already underway. The newly proposed biological definition of PD emphasizes that such changes can be detected via appropriate biomarkers before the onset of clinical manifestations [1,2].
MRI may play a critical role in identifying individuals in this stage, particularly those with recognized risk factors such as RBD, anosmia, depression, or LRRK2 and GBA1 mutations.
Early changes in SNpc and striatum
Population-based studies have shown that individuals with isolated RBD (iRBD) – a strong prodromal marker of PD – exhibit a reduced neuromelanin signal in the SNpc and increased magnetic susceptibility on QSM, similar to findings observed in patients with manifest PD [20].
In the study by Schwarz et al. [16], individuals with iRBD exhibited reduced SNpc volume on NM-MRI and signs of N1 degeneration, suggesting the possibility of detecting significant structural abnormalities before the appearance of tremor or rigidity.
Connectomics and network alterations
Advanced fMRI techniques and connectome analyses using both structural and fMRI data allow for identification of alterations within cortico-basal and limbic networks. Basaia et al. [21] developed a predictive model based on 3T MRI data from PD patients and healthy controls, which accurately identified regions at risk for future gray matter atrophy even at early disease stages.
Such functional alterations – including reduced connectivity in the executive and default mode networks – are also observed in patients with mild cognitive impairment or depression, suggesting their potential utility as early prodromal markers [19].
MRI correlations with positron emission tomography (PET) and other biomarkers
While MRI does not provide direct measures of dopaminergic activity like PET or dopamine transporter single photon emission computed tomography (DAT-SPECT), increasing evidence supports the correlation between MRI-based structural and functional biomarkers and molecular imaging findings. For instance, reduced NM-MRI signal in the SNpc has been shown to correlate with decreased DAT uptake in PET and SPECT, highlighting the potential of NM-MRI as a surrogate marker for molecular pathology [22].
In the near future, MRI may become an integral part of multimodal screening protocols, particularly in genetically or clinically at-risk populations.
The role of artificial intelligence (AI) and radiomics in MRI analysis
The rapid advancement of AI, particularly in the domains of machine learning and deep learning, has created new opportunities for the analysis of MRI in PD. One of the most promising applications is radiomics – the automated extraction of quantitative features from medical images – which enables the detection of subtle imaging patterns that may be imperceptible to the human eye yet correlate with neurodegenerative processes.
Recent studies have demonstrated the potential of radiomics and AI not only for diagnostic purposes but also for risk stratification and progression prediction, especially in the prodromal stages of PD.
Radiomics applied to routine MRI sequences
In a landmark 2025 study, Ye et al. [23] applied radiomic algorithms to routine T2W FLAIR MRI scans in a large cohort of subjects. The resulting classification models achieved an AUC ranging from 0.85 to 0.98 and diagnostic accuracy between 78% and 90% in differentiating PD patients from healthy controls.
Notably, radiomic models based on FLAIR sequences captured white matter and subcortical alterations that correlated with early clinical signs. These findings suggest a potential role for radiomics in population-level screening and early classification of individuals at risk of developing PD.
Predictive modeling and deep neural networks
Deep convolutional neural networks have enabled high-accuracy classification of MRI data. In recent years, several multimodal models have emerged that integrate morphometric, DTI, and fMRI data with clinical metrics such as the Unified Parkinson’s Disease Rating Scale. These models have demonstrated high predictive accuracy for disease progression [24].
Some deep learning frameworks have also been trained to forecast conversion from prodromal stages – such as iRBD – to clinically manifest PD by analyzing connectivity patterns and early gray matter atrophy [25].
Limitations and implementation challenges
Despite their potential, the clinical translation of AI-based algorithms for PD diagnosis remains limited by several practical and methodological constraints:
lack of input data standardization, including differences in MRI acquisition protocols, scanner models, and sequence parameters;
limited availability of high-quality reference-labeled datasets, particularly those validated with PET, DAT-SPECT, or histopathology;
insufficient external validation and a shortage of independent multicenter studies;
skepticism among clinicians, often due to the “black-box” nature of AI decision-making.
Additionally, the radiologist’s expertise remains critical in interpreting AI-generated outputs. Studies support a human-in-the-loop approach, in which algorithmic predictions are reviewed and verified by an experienced specialist, achieving the best diagnostic synergy [15].
MRI in the differential diagnosis of PD and atypical parkinsonian syndromes
Differentiating idiopathic PD from atypical parkinsonian syndromes – such as MSA, PSP, corticobasal degeneration (CBD), and dementia with Lewy bodies (DLB) – remains a major diagnostic challenge, particularly in early stages of disease. Conventional clinical assessment is often insufficient, as overlapping motor features (bradykinesia, rigidity, tremor) may delay accurate diagnosis for several years. MRI has emerged as a key noninvasive modality in identifying characteristic structural changes and distribution patterns of neurodegeneration that aid in differential diagnosis.
Key MRI features in atypical syndromes
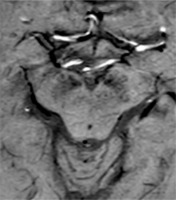
In MSA, MRI typically reveals atrophy of the cerebellum, brainstem, and putamen, as well as characteristic signal abnormalities. The so-called “hot cross bun sign” in the pons and hyperintense lateral putaminal rims on T2W images are highly suggestive of MSA-C and MSA-P phenotypes, respectively [26] (Figures 2 and 3).
Figure 2
A, B, C, D Midbrain morphometry. MRI axial FLAIR A) Line expresses the transverse diameter of the midbrain at the the level of the superior colliculus B) The thickness of the superior cerebellar peduncle at the level of the inferior colliculus C) Thickness of the inferior cerebellar peduncle D) Transverse diameter of the medulla oblongata at the level of the inferior olivary nucleus
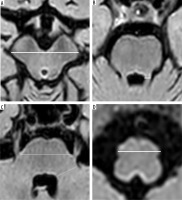
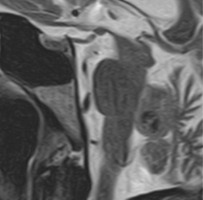
Figure 3
Measuring the pons and midbrain area. FLAIR sagittal image. Elliptical regions of interest were placed over the pons and the midbrain in the midsagittal slice
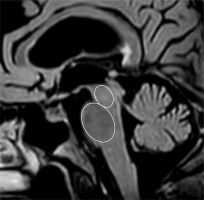
In PSP, midbrain atrophy with preserved pons volume (leading to the “hummingbird sign” or “morning glory sign” in sagittal view) is a classic imaging finding (Figure 4). Volumetric studies have identified reductions in midbrain tegmental area and superior cerebellar peduncles as robust MRI biomarkers [27].
CBD typically presents with asymmetric cortical atrophy, predominantly involving the frontoparietal cortex, with additional subcortical volume loss. MRI findings in CBD are less specific and may overlap with other disorders; however, patterns of regional atrophy support the clinical diagnosis [28].
In DLB, medial temporal lobe atrophy is usually mild compared to that seen in Alzheimer’s disease. DLB may also show occipital hypoperfusion or hypometabolism, though these features are more reliably detected with PET or SPECT than with MRI [29].
The role of quantitative and advanced MRI
Advanced MRI techniques such as volumetry, DTI, and QSM have shown promise in refining the diagnostic differentiation. For example, diffusion metrics in the superior cerebellar peduncles and midbrain show consistent abnormalities in PSP, while increased magnetic susceptibility in the putamen is characteristic of MSA. The integration of these quantitative biomarkers improves diagnostic sensitivity and specificity compared to visual inspection alone [30].
Neuromelanin imaging and N1 visualization are useful in distinguishing PD from non-degenerative mimics (e.g., essential tremor), but offer limited utility in differentiating PD from atypical parkinsonian syndromes, where degeneration patterns extend beyond the SN.
Clinical implications and limitations
Although MRI contributes significantly to the diagnostic process, no single imaging marker currently allows for definitive differentiation of all parkinsonian syndromes. Multimodal imaging protocols that combine conventional sequences, iron-sensitive imaging, and diffusion metrics –interpreted within the clinical context – offer the best diagnostic accuracy.
Moreover, age-specific considerations are important: in younger patients (< 50 years), atypical parkinsonism is rare, and MRI is most useful in excluding structural or metabolic causes. In contrast, in elderly patients, where vascular parkinsonism and atypical syndromes are more prevalent, MRI should be supplemented with nuclear imaging e.g., DAT-SPECT or fluorodeoxyglucose PET (FDG-PET), to clarify etiology (Table 2).
Table 2
Recommended magnetic resonance imaging (MRI) protocols and complementary diagnostic tests in patients with suspected Parkinson’s disease, stratified by age group
[i] AI – artificial intelligence, CBD – corticobasal degeneration, CSF – cerebrospinal fluid, DAT-SPECT – dopamine transporter single-photon emission computed tomography, DTI – diffusion tensor imaging, FDG-PET – fluorodeoxyglucose positron emission tomography, FDOPA – fluorodopa, FLAIR – fluid-attenuated inversion recovery, fMRI – functional magnetic resonance imaging, MRPI – magnetic resonance parkinsonism index, MSA – multiple system atrophy, NM-MRI – neuromelanin-sensitive magnetic resonance imaging, PET – positron emission tomography, PSP – progressive supranuclear palsy, QSM – quantitative susceptibility mapping, SWI – susceptibility-weighted imaging
Limitations of MRI and complementary imaging techniques
Despite its growing importance in the diagnostic and research domains of PD, MRI still faces several limitations that constrain its clinical utility. While MRI enables visualization of structural and functional changes associated with neurodegeneration, it does not directly reflect the molecular hallmarks of PD, such as α-synuclein accumulation or dopaminergic dysfunction.
First, MRI is inherently limited by its indirect nature. Even advanced techniques – such as NM-MRI, SWI, QSM, and DTI – provide only surrogate markers of neurodegeneration, based on structural or microstructural tissue alterations. They do not capture synaptic activity, dopamine transporter loss, or protein aggregates, which are better assessed by nuclear imaging techniques. Moreover, standard anatomical MRI sequences (T1W, T2W, and FLAIR) lack specificity and are typically used to exclude secondary causes of parkinsonism (e.g., vascular, neoplastic, or normal pressure hydrocephalus).
Second, the availability and technical variability of high-resolution and ultra-high-field MRI (especially 7T) pose major challenges. Although 7T MRI allows unprecedented spatial resolution and is highly effective in visualizing N1 degeneration, its widespread clinical use is hindered by cost, limited access, and the lack of standardized acquisition protocols. Field strength, coil configuration, and scanner vendor introduce significant heterogeneity in image quality and contrast, limiting reproducibility across centers.
Another important factor is the dependence on radiologist experience. Interpretation of NM-MRI and iron-sensitive sequences is highly operator-dependent. Studies have demonstrated that radiologists with dedicated training in neurodegeneration achieve substantially higher diagnostic accuracy than general radiologists, particularly in tasks such as identifying the “swallow tail” sign or quantifying signal loss in the SN. In the absence of structured interpretation guidelines and normative atlases, diagnostic performance remains inconsistent, especially in early or prodromal PD.
To address these limitations, MRI is increasingly combined with complementary imaging modalities. Dopamine transporter single-photon emission computed tomography (DAT-SPECT) is a widely used tool for assessing presynaptic dopaminergic integrity and is especially helpful in early disease stages when MRI findings are subtle or equivocal. Similarly, fluorine-18 FDG-PET can aid in differentiating PD from atypical parkinsonian syndromes such as MSA or PSP by revealing characteristic metabolic patterns. The emergence of PET tracers targeting synaptic vesicle proteins (e.g., SV2A) and pathological proteins (e.g., α-synuclein, tau) also opens the door to biologically grounded diagnostic protocols, though these techniques are currently restricted to research settings.
From a clinical perspective, age- and phenotype-specific diagnostic strategies should be considered. In young-onset PD (< 50 years), MRI serves not only to assess structural integrity but also to rule out genetic, metabolic, or inflammatory etiologies (e.g., Wilson’s disease, leukodystrophies, mitochondrial disorders). In this subgroup, the threshold for extended diagnostics – including cerebrospinal fluid analysis and genetic testing – is significantly lower. Conversely, in older adults, where vascular parkinsonism, atypical syndromes, or mixed pathologies are more common, structural MRI should be supplemented with DAT-SPECT or PET to increase diagnostic accuracy and guide clinical management.
In summary, while MRI represents a powerful and evolving tool for the evaluation of PD, its limitations must be acknowledged. The integration of MRI with molecular imaging, clinical phenotyping, and, in select cases, genetic and biomarker analysis constitutes the most comprehensive and reliable approach to the modern diagnostic pathway in PD.
Conclusions and future perspectives
MRI is increasingly recognized as a central tool in the evolving diagnostic landscape of PD. While historically considered non-specific in PD diagnosis, contemporary techniques – such as NM-MRI, susceptibility-based imaging, high-field MRI, and radiomics – have markedly expanded the scope of MRI beyond structural exclusion toward direct assessment of disease-specific neurodegenerative changes.
Recent shifts toward a biological definition of PD, as proposed by the International Parkinson and Movement Disorder Society and other expert groups, emphasize the need for objective, non-invasive biomarkers that can identify pathological changes in the prodromal and preclinical phases of disease. MRI is well positioned to fulfil this role, particularly when used in conjunction with molecular imaging and clinical data. Techniques such as 7T neuromelanin imaging and connectome-based modelling now allow for increasingly precise mapping of early degeneration, while radiomic and AI-based tools hold promise for scalable, automated screening approaches.
Nonetheless, important limitations persist. MRI remains an indirect measure of pathology, and interpretation still relies heavily on radiologist expertise, scanner availability, and institutional experience. Standardization of protocols, multicenter validation of imaging biomarkers, and integration into diagnostic algorithms endorsed by international guidelines are essential next steps.
From a clinical perspective, MRI should be used not only for differential diagnosis – particularly in distinguishing idiopathic PD from atypical parkinsonian syndromes – but also as a biomarker of progression and treatment response, especially in the context of clinical trials. For younger patients, comprehensive MRI combined with genetic and metabolic workup remains crucial. In older adults, multimodal imaging strategies that incorporate MRI with PET or SPECT offer the best diagnostic yield.
In conclusion, MRI is evolving from a supportive tool to a potential biomarker platform in PD, aligning with the emerging emphasis on biological staging and personalized medicine. Further research into harmonized imaging protocols, combined biomarker models, and AI integration will be pivotal in transitioning from research settings into routine clinical application.