Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has rapidly spread across the globe, leading to a pandemic known as COVID-19. Since December 2019, it has affected 770 million people, resulting in over 6.9 million deaths (according to data from the WHO as of 18 July 2022) [1]. An unpredictable, rapidly spreading disease with many fatalities has caused mass hysteria, economic burden, and financial losses [2]. Nonspecific symptoms, easy transmission of the pathogen, and initial diagnostic challenges have contributed to the rapid spread of the pandemic and an unprecedented global public health crisis [3].
A real-time reverse transcription-polymerase chain reaction (RT-PCR), due to its high specificity, is considered the gold standard for COVID-19 detection. However, false negative results can occur due to factors such as the quality and the type of the specimen, how it is processed, and the stage of infection when the sample is collected [4,5]. In cases of false-negative RT-PCR results and worsening clinical symptoms, imaging becomes critical for patients suspected of having COVID-19 [5]. During the early stages of the pandemic, due to RT-PCR tests shortages, chest X-rays (CXR) and chest computed tomography (CT) were widely used as a part of the initial screening process [5,6]. Furthermore, some authors proposed chest CT as the modality able to rectify false negatives obtained from RT-PCR during the early stages of disease [7,8]. Given its comprehensive insights, chest imaging has played various roles during the pandemic, including diagnosing, triaging, and assessing the prognosis of patients with COVID-19 pneumonia [9-11]. Although not routinely indicated as the screening test in asymptomatic patients, CXR remains the first-line imaging modality for patients with COVID-19 [5,12].
Despite being less sensitive than RT-PCR and CT [13], CXR are a quick, cost-effective, and widely available imaging modality, providing sufficient lung assessment in the majority of COVID-19 cases [12]. Despite the typically normal CXR of early and mildly symptomatic patients, and some discrepancies among authors describing changes typical for COVID-19 on CXR, it is possible to distinguish a number of symptoms typical for this disease [14]. Consolidations, ground glass opacities, bilateral involvement, and peripheral distribution of changes are the basis for diagnosing COVID-19 on CXR as well as in CT [13,14]. Given the significant increase in the number of radiological examinations during the pandemic, it has become necessary to explore methods of automatic image analysis. This would allow for an increase in the diagnostic parameters of routine CXR in detecting COVID-19. Recent research has demonstrated that artificial intelligence (AI), convolution neural network (CNN), and image texture analysis can be useful tools to assist radio-logists in various clinical scenarios, including COVID-19 detection and evaluation [15-18]. However, despite the many studies conducted, the optimal tools and their impact on patient care and outcomes are still uncertain [19]. Therefore, further research into the image texture parameters of COVID-19 patients is essential.
The purpose of this study was to compare the results of CXR texture analysis in patients suspected of COVID-19 based on a subjective radiologist’s opinion with a control group of healthy individuals who had no abnormal findings on their CXR. All the patients from the study group had a RT-PCR-confirmed COVID-19 diagnosis.
Material and methods
The study was retrospective and therefore did not require approval from a bioethical committee. The study material consisted of CXRs performed on patients admitted to our institution. The inclusion criteria were as follows: (1) confirmation of SARS-CoV-2 infection by RT-PCR, (2) clinical suspicion of COVID-19 pneumonia, and (3) a technically adequate CXR in the postero-anterior projection. All the inclusion criteria had to be met for inclusion in the study group.
The exclusion criteria included severe image motion artifacts and low overall image quality due to inadequate exposition parameters. The control group consisted of 30 CXRs of different patients with no abnormal findings in CXR and without SARS-CoV-2 confirmation.
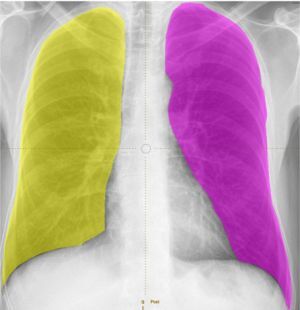
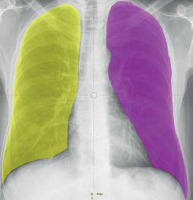
Radiographs were taken on a DR7500 unit (Care-stream Health, Inc., Rochester, NY, USA). Image texture parameters were analysed with LIFEx software using DICOM files [20,21]. The following initial assumptions were made: 128 shades of grey tested, and the region of interest (ROI) separate for each lung (Figure 1). All the available texture parameters were analysed. A result was a positive diagnosis of COVID-19 pneumonia in the test group vs. control group. Results included only significant parameters.
The results were statistically analysed using MedCalc software (v. 22.016, Ostend, Belgium). The ROC curve was used to assess the diagnostic value of the consecutive radiomics variables. Stepwise logistic regression analysis was used to find the best model to predict COVID-19-related pneumonia. The significance level for all statistical tests was set to 0.05.
Results
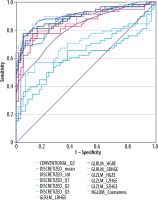
The study group consisted of 70 patients (46 males, 24 females) with a mean age of 52 years. The control group consisted of 30 selected patients (21 males, 9 females) with a mean age of 53 years. The control group was selected to match the study group by age. Of the 57 texture parameters tested, 13 presented a statistically significant power to distinguish between COVID-19 pneumonia and a healthy subject (Table 1). The highest AU presented GLRLM_LRHGE (0.91). All the relevant ROC curves are shown in Figure 2. Stepwise logistic regression analysis found a model with a significance level of p < 0.0001. The included variables were DISCRETIZED_Q3 and DISCRETIZED_std (Table 2). The model presented an AUC of 0.95 (95% CI: 0.90-0.98).
Table 1
Statistically significant image texture parameters for discriminating between patients with COVID-19 pneumonia and healthy subjects
Discussion
Advanced computer analysis of radiological images (e.g. artificial intelligence) was intended to improve the workflow in radiology. One of those methods, i.e. texture ana-lysis, is an attempt to extract more information from imaging than conventionally possible. This was particularly important during the COVID-19 pandemic, when the number of CXRs increased rapidly and when other chest pathologies become “less important”. Sadly, we entered a situation in which CXR was for some time used just to discriminate between COVID-19 pneumonia and other conditions. Of course, this was a simplified version of the situation, but it was forced by the pandemic. The present study showed that chest X-ray image texture analysis is an easy and accurate supportive tool for discriminating between COVID-19 pneumonia and healthy subjects. With all the limitations, our results encourage further investigations on the use of texture analysis in imaging.
To date, there is no consensus on who should bear responsibility for medical and diagnostic decision-making when AI is involved. While clinicians are ultimately responsible for making medical decisions, the use of AI algorithms complicates the picture because the authors of the algorithms are usually not medically trained and may not fully understand the implications of their algorithms on medical decision-making. This raises important ethical issues related to accountability and transparency in medical decision-making. Medical decisions made using AI algorithms must be transparent, and the process by which the algorithm makes its recommendations must be understandable to both medical professionals and patients. Furthermore, there must be clear guidelines for who takes responsibility in cases where medical decisions are made using AI algorithms.
The most frequent findings related to COVID-19 on chest radiographs are ground-glass opacities, followed by diffuse air space disease, bilateral lower lobe consolidations, and peripheral air space opacities. Pleural effusions, lung cavitation, and pneumothorax may occur, but they can be considered as bacterial complications of the primary viral infection [4,16,19]. There have been several studies testing the use of computer-aided diagnosis of COVID-19 pneumonia on CXR. For instance, in a study by Murphy et al. [16] the results of AI readings, compared to a group of radiologists, were encouraging, with an AUC of 0.81. However, the results strongly relate to the clinical advancement of the disease. The appearance of COVID-19 pneumonia on CXR can be highly variable, ranging from peripheral opacifications only to diffuse opacifications, which makes differentiation from other diseases challenging [4,18,19]. In our study, patients were scanned at the level of admission, which might had reduced the percentage of cases of more advanced pneumonia. Chest radiographs may be normal initially or may show mild disease that is difficult to diagnose by both radiologist and TA [16]. However, Wong et al. [13] showed that of all the patients with COVID-19 who required hospitalization, 69% had abnormal chest radiograph findings at admission. During hospitalization, 80% showed chest radiograph abnormalities, which were most extensive 10-12 days after symptom onset [13].
Our study had several limitations that should be addressed. First, the sample size was relatively small, and our preliminary results must be validated on much larger sample. Nowadays, the SARS-CoV-2 pandemic tends to decrease and present fewer pneumonia symptoms. The question is that if it ends, will a new viral infection that requires intensive imaging then explode. Second, the choice of ROI was important for the study. We adopted a specific criterion of the lung outline using the ROI; however, for diseased lungs, determining the entire lung space using the ROI is more difficult than for the image of healthy lungs, in which the lung tissue is clearly distinguishable from the other tissues. Third, the analysis included only healthy/diseased dichotomization that did not take into account lung pathologies other than COVID-19. Patients with chronic interstitial lung diseases with SARS-CoV-2 infection might in particular be a source of false positive cases. Finally, the protocol of the study could have hidden SARS-CoV-2-positive subjects with previous interstitial lung diseases.
Conclusions
Selected texture parameters make it possible to distinguish images of lungs affected by COVID-19 lesions from healthy ones. In future studies, the role of AI in the care or triage of patients during the COVID-19 pandemic should be investigated, taking all related patient information and the experience level of the health care professionals interpreting the radiographs into account.