UROGENITAL RADIOLOGY / ORIGINAL PAPER
Multiphase CT-derived markers for the characterization of large solid and cystic renal masses by histology and grade
1
Voxel Medical Diagnostic Center, Katowice, Poland
2
Danylo Halytsky Lviv National Medical University, Lviv, Ukraine
Submission date: 2025-07-03
Final revision date: 2025-08-10
Acceptance date: 2025-11-03
Publication date: 2026-03-02
Corresponding author
Pol J Radiol, 2026; 91(1): 95-114
KEYWORDS
renal cell carcinomaradiomicsimaging markermultiphase contrast-enhanced CTlarge renal massescystic renal cell carcinoma
TOPICS
ABSTRACT
Purpose:
This study aimed to evaluate the diagnostic performance of multiphase contrast-enhanced computed tomography (MCECT) for differentiating benign and malignant renal masses > 4 cm, and in predicting renal cell carcinoma (RCC) subtypes and grade, using signal intensity (SI) and tumor-to-cortex signal intensity ratio (TCSI).
Material and methods:
A retrospective analysis was performed on 190 patients with renal tumors > 4 cm (120 solid and 70 cystic lesions). All solid tumors and 49 cystic lesions (Bosniak IIF-IV) underwent histopathological verification. MCECT was performed in four phases: pre-contrast, corticomedullary (CMP), nephrographic (NP), and excretory (EP). SI and TCSI were measured and analyzed using receiver operating characteristic analysis. Cluster and principal component analyses were applied to evaluate enhancement-based classification.
Results:
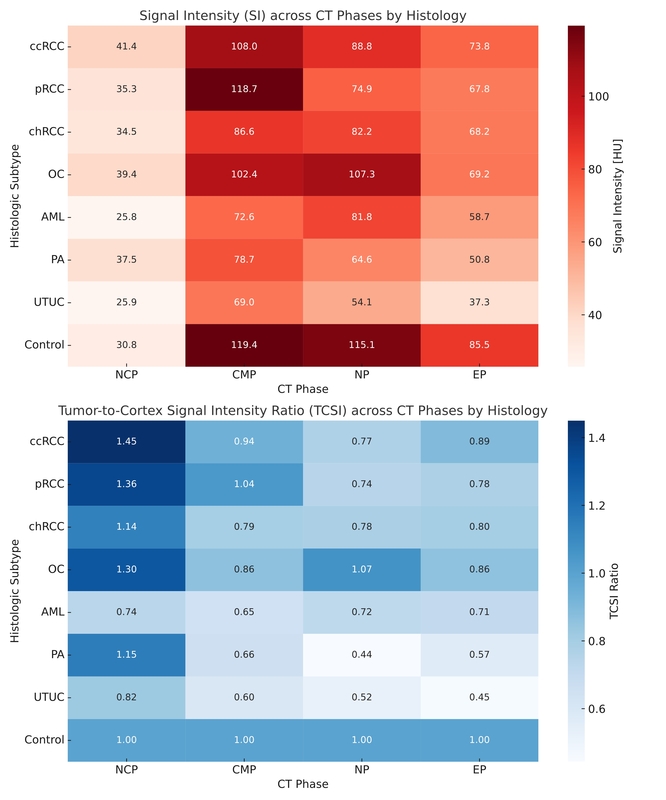
For solid masses, excretory phase SI reached an area under the curve (AUC) of 0.844 for distinguishing RCC from other tumors (98.8% sensitivity, 69.4% specificity). Differentiating RCC from benign tumors using EP SI achieved an AUC of 0.745. CMP SI enabled separation of RCC subtypes, especially chRCC vs. pRCC (AUC = 0.983). SI of NP differentiated low- from high-grade ccRCC with an AUC of 0.969 (100% sensitivity, 90.9% specificity). Among Bosniak IIF cysts, EP TCSI ≥ 0.40 identified malignancy with 88.9% sensitivity and 100% specificity (AUC = 0.951). Cluster analysis grouped tumors by vascularity-based enhancement. Lymph node assessment showed no significant SI differences between pN1 and pN0.
Conclusions:
SI and TCSI from MCECT are accurate, non-invasive markers for histologic and biologic characterization of large renal masses. CMP and EP provide the highest diagnostic value. These enhancement parameters may improve radiologic workflows and support clinical decision-making.
This study aimed to evaluate the diagnostic performance of multiphase contrast-enhanced computed tomography (MCECT) for differentiating benign and malignant renal masses > 4 cm, and in predicting renal cell carcinoma (RCC) subtypes and grade, using signal intensity (SI) and tumor-to-cortex signal intensity ratio (TCSI).
Material and methods:
A retrospective analysis was performed on 190 patients with renal tumors > 4 cm (120 solid and 70 cystic lesions). All solid tumors and 49 cystic lesions (Bosniak IIF-IV) underwent histopathological verification. MCECT was performed in four phases: pre-contrast, corticomedullary (CMP), nephrographic (NP), and excretory (EP). SI and TCSI were measured and analyzed using receiver operating characteristic analysis. Cluster and principal component analyses were applied to evaluate enhancement-based classification.
Results:
For solid masses, excretory phase SI reached an area under the curve (AUC) of 0.844 for distinguishing RCC from other tumors (98.8% sensitivity, 69.4% specificity). Differentiating RCC from benign tumors using EP SI achieved an AUC of 0.745. CMP SI enabled separation of RCC subtypes, especially chRCC vs. pRCC (AUC = 0.983). SI of NP differentiated low- from high-grade ccRCC with an AUC of 0.969 (100% sensitivity, 90.9% specificity). Among Bosniak IIF cysts, EP TCSI ≥ 0.40 identified malignancy with 88.9% sensitivity and 100% specificity (AUC = 0.951). Cluster analysis grouped tumors by vascularity-based enhancement. Lymph node assessment showed no significant SI differences between pN1 and pN0.
Conclusions:
SI and TCSI from MCECT are accurate, non-invasive markers for histologic and biologic characterization of large renal masses. CMP and EP provide the highest diagnostic value. These enhancement parameters may improve radiologic workflows and support clinical decision-making.
REFERENCES (41)
1.
Howlader N, Noone A, Krapcho M, Miller D, Brest A, Yu M, et al. SEER cancer statistics review, 1975-2017. National Cancer Institute. Bethesda, MD; 2020. Available from: https://seer.cancer.gov/csr/19....
2.
Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2024; 74: 229-63.
3.
Bex A, Ghanem YA, Albiges L, Bonn S, Campi R, Capitanio U, et al. European Association of Urology guidelines on renal cell carcinoma: the 2025 update. Eur Urol 2025; 87: 683-696.
4.
Blute M, Prince J, Bultman E, Hinshaw L, Drewry A, Best S, et al. MP44-16 predictors of non-diagnostic renal mass biopsy. J Urol 2015; 193: e532-533.
5.
Marconi L, Dabestani S, Lam TB, Hofmann F, Stewart F, Norrie J, et al. Systematic review and meta-analysis of diagnostic accuracy of percutaneous renal tumour biopsy. Eur Urol 2016; 69: 660-673.
6.
Corcoran AT, Russo P, Lowrance WT, Asnis-Alibozek A, Libertino JA, Pryma DA, et al. A review of contemporary data on surgically resected renal masses-benign or malignant? Urology 2013; 81: 707-713.
7.
Ishigami K, Jones AR, Dahmoush L, Leite LV, Pakalniskis MG, Barloon TJ. Imaging spectrum of renal oncocytomas: a pictorial review with pathologic correlation. Insights Imaging 2014; 6: 53-64.
8.
Defortescu G, Cornu JN, Béjar S, Giwerc A, Gobet F, Werquin C, et al. Diagnostic performance of contrast-enhanced ultrasonography and magnetic resonance imaging for the assessment of complex renal cysts: a prospective study. Int J Urol 2017; 24: 184-189.
9.
Parihar AS, Mhlanga J, Ronstrom C, Schmidt LR, Figenshau RS, Dehdashti F, et al. Diagnostic accuracy of 99mTc-Sestamibi SPECT/CT for characterization of solid renal masses. J Nucl Med 2023; 64: 90-95.
10.
Singhal T, Singh P, Parida GK, Agrawal K. Role of PSMA-targeted PET-CT in renal cell carcinoma: a systematic review and meta-analysis. Ann Nucl Med 2024; 38: 176-187.
11.
Fujii Y, Komai Y, Saito K, Iimura Y, Yonese J, Kawakami S, et al. Incidence of benign pathologic lesions at partial nephrectomy for presumed RCC renal masses: Japanese dual-center experience with 176 consecutive patients. Urology 2008; 72: 598-602.
12.
Fujii Y, Saito K, Iimura Y, Yasuda Y, Koga F, Masuda H, et al. Incidence of benign pathologic lesions at nephrectomy for renal masses presumed to be stage I renal cell carcinoma in Japanese patients: impact of sex, age, and tumor size. ASCO Meeting Abstracts 2011; 29: 374.
13.
Vijay V, Vokshi FH, Smigelski M, Nagpal S, Huang WC. Incidence of benign renal masses in a contemporary cohort of patients receiving partial nephrectomy for presumed renal cell carcinoma. Clin Genitourin Cancer 2023; 21: e114-e118.
14.
van den Brink L, Debelle T, Gietelink L, Graafland N, Ruiter A, Bex A, et al. A national study of the rate of benign pathology after partial nephrectomy for T1 renal cell carcinoma: should we be satisfied? Cancers (Basel) 2024; 16: 3518.
15.
Dehghani Firouzabadi F, Gopal N, Hasani A, Homayounieh F, Li X, Jones EC, et al. CT radiomics for differentiating fat poor angiomyolipoma from clear cell renal cell carcinoma: systematic review and meta-analysis. PLoS One 2023; 18: e0287299.
16.
Kan HC, Lin PH, Shao IH, Cheng SC, Fan TY, Chang YH, et al. Using deep learning to differentiate among histology renal tumor types in computed tomography scans. BMC Med Imaging 2025; 25: 66.
17.
Coy H, Young JR, Douek ML, Brown MS, Sayre J, Raman SS. Quantitative computer-aided diagnostic algorithm for automated detection of peak lesion attenuation in differentiating clear cell from papillary and chromophobe renal cell carcinoma, oncocytoma, and fat-poor angiomyolipoma on multiphasic multidetector computed tomography. Abdom Radiol (NY) 2017; 42: 1919-1928.
18.
Coy H, Hsieh K, Wu W, Nagarajan MB, Young JR, Douek ML, et al. Deep learning and radiomics: the utility of Google TensorFlowTM Inception in classifying clear cell renal cell carcinoma and oncocytoma on multiphasic CT. Abdom Radiol (NY) 2019; 44: 2009-2020.
19.
Ma Y, Ma W, Xu X, Guan Z, Pang P. A convention-radiomics CT nomogram for differentiating fat-poor angiomyolipoma from clear cell renal cell carcinoma. Sci Rep 2021; 11: 4644.
20.
Wei J, Ma Y, Liu J, Zhao J, Zhou J. A noninvasive comprehensive model based on medium sample size had good diagnostic performance in distinguishing renal fat-poor angiomyolipoma from homogeneous clear cell renal cell carcinoma. Urol Oncol 2025; 43: 332.e1-332.e10.
21.
Mytsyk Y. Differentiation of solid and cystic small renal masses: the role of multiphase CT markers in predicting malignant histology, subtype, and grade. Pol J Radiol 2025; 90: 239-252.
22.
Silverman SG, Pedrosa I, Ellis JH, Hindman NM, Schieda N, Smith AD, et al. Bosniak Classification of Cystic Renal Masses, Version 2019: an update proposal and needs assessment. Radiology 2019; 292: 475-488.
23.
Mytsyk Y, Dutka I, Yuriy B, Maksymovych I, Caprnda M, Gazdikova K, et al. Differential diagnosis of the small renal masses: role of the apparent diffusion coefficient of the diffusion-weighted MRI. Int Urol Nephrol 2018; 50: 197-204.
24.
Mytsyk Y, Pasichnyk S, Dutka I, Dats I, Vorobets D, Skrzypczyk M, et al. Systemic treatment of the metastatic renal cell carcinoma: usefulness of the apparent diffusion coefficient of diffusion-weighted MRI in prediction of early therapeutic response. Clin Exp Med 2020; 20: 277-287.
25.
Mytsyk Y, Borzhiyevskyy A, Dutka I, Shulyak A, Kowal P, Vorobets D, et al. Local recurrence of renal cell carcinoma after partial nephrectomy: applicability of the apparent diffusion coefficient of MRI as an imaging marker – a multicentre study. Pol J Radiol 2022; 87: 325-332.
26.
Kowal P, Ratajczyk K, Bursiewicz W, Trzciniecki M, Marek-Bukowiec K, Rogala J, et al. Differentiation of solid and friable tumour thrombus in patients with renal cell carcinoma: the role of MRI apparent diffusion coefficient. Adv Med Sci 2024; 69: 434-442.
27.
Young JR, Margolis D, Sauk S, Pantuck AJ, Sayre J, Raman SS. Clear cell renal cell carcinoma: discrimination from other renal cell carcinoma subtypes and oncocytoma at multiphasic multidetector CT. Radiology 2013; 267: 444-453.
28.
Kim SH, Kim CS, Kim MJ, Cho JY, Cho SH. Differentiation of clear cell renal cell carcinoma from other subtypes and fat-poor angiomyolipoma by use of quantitative enhancement measurement during three-phase MDCT. AJR Am J Roentgenol 2016; 206: W21-28.
29.
Trevisani F, Floris M, Minnei R, Cinque A. Renal oncocytoma: the diagnostic challenge to unmask the double of renal cancer. Int J Mol Sci 2022; 23: 2603.
30.
Yang G, Gong A, Nie P, Yan L, Miao W, Zhao Y, et al. Contrast-enhanced CT texture analysis for distinguishing fat-poor renal angiomyolipoma from chromophobe renal cell carcinoma. Mol Imaging 2019; 18: 1536012119883161.
31.
He Z, Liu H, Moch H, Simon HU. Machine learning with autophagy-related proteins for discriminating renal cell carcinoma subtypes. Sci Rep 2020; 10: 720.
32.
Dehghani Firouzabadi F, Gopal N, Homayounieh F, Anari PY, Li X, Ball MW, et al. CT radiomics for differentiating oncocytoma from renal cell carcinomas: systematic review and meta-analysis. Clin Imaging 2023; 94: 9-17.
33.
Kocak B, Yardimci AH, Bektas CT, Turkcanoglu MH, Erdim C, Yucetas U, et al. Textural differences between renal cell carcinoma subtypes: machine learning-based quantitative computed tomography texture analysis with independent external validation. Eur J Radiol 2018; 107: 149-157.
34.
Mühlbauer J, Egen L, Kowalewski KF, Grilli M, Walach MT, Westhoff N, et al. Radiomics in renal cell carcinoma – a systematic review and meta-analysis. Cancers (Basel) 2021; 13: 1348.
35.
Lucocq J, Pillai S, Oparka R, Nabi G. Complex renal cysts (Bosniak ≥ IIF): interobserver agreement, progression and malignancy rates. Eur Radiol 2021; 31: 901-908.
36.
Pruthi DK, Liu Q, Kirkpatrick IDC, Gelfond J, Drachenberg DE. Long-term surveillance of complex cystic renal masses and heterogeneity of Bosniak 3 lesions. J Urol 2018; 200: 1192-1199.
37.
Wang T, Yang H, Hao D, Nie P, Liu Y, Huang C, et al. A CT-based radiomics nomogram for distinguishing between malignant and benign Bosniak IIF masses: a two-centre study. Clin Radiol 2023; 78: 590-600.
38.
Zhao X, Yan Y, Xie W, Qin Z, Zhao L, Liu C, et al. Radiomics for differential diagnosis of Bosniak II-IV renal masses via CT imaging. BMC Cancer 2024; 24: 1508.
39.
Yin Y, Campbell SP, Markowski MC, Pierorazio PM, Pomper MG, Allaf ME, et al. Inconsistent detection of sites of metastatic non-clear cell renal cell carcinoma with PSMA-targeted [18F]DCFPyL PET/CT. Mol Imaging Biol 2019; 21: 567-573.
40.
Tadayoni A, Paschall AK, Malayeri AA. Assessing lymph node status in patients with kidney cancer. Transl Androl Urol 2018; 7: 766-773.
41.
Xiong M, Zhang W, Zhou C, Bao J, Zang S, Lin X. Application of 18F prostate-specific membrane antigen positron emission tomography/computed tomography in monitoring gastric metastasis and cancer thrombi from renal cell carcinoma. J Oncol 2022; 2022: 5681463.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.